Capture, mutual inhibition and release mechanism for aPKC–Par6 and its multisite polarity substrate Lgl
IF 10.1
1区 生物学
Q1 BIOCHEMISTRY & MOLECULAR BIOLOGY
引用次数: 0
Abstract
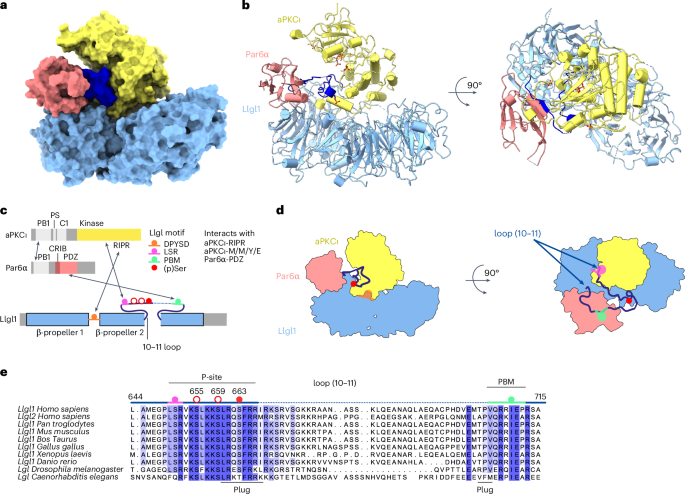
The mutually antagonistic relationship of atypical protein kinase C (aPKC) and partitioning-defective protein 6 (Par6) with the substrate lethal (2) giant larvae (Lgl) is essential for regulating polarity across many cell types. Although aPKC–Par6 phosphorylates Lgl at three serine sites to exclude it from the apical domain, aPKC–Par6 and Lgl paradoxically form a stable kinase–substrate complex, with conflicting roles proposed for Par6. We report the structure of human aPKCι–Par6α bound to full-length Llgl1, captured through an aPKCι docking site and a Par6PDZ contact. This complex traps a phospho-S663 Llgl1 intermediate bridging between aPKC and Par6, impeding phosphorylation progression. Thus, aPKCι is effectively inhibited by Llgl1pS663 while Llgl1 is captured by aPKCι–Par6. Mutational disruption of the Lgl–aPKC interaction impedes complex assembly and Lgl phosphorylation, whereas disrupting the Lgl–Par6PDZ contact promotes complex dissociation and Lgl phosphorylation. We demonstrate a Par6PDZ-regulated substrate capture-and-release model requiring binding by active Cdc42 and the apical partner Crumbs to drive complex disassembly. Our results suggest a mechanism for mutual regulation and spatial control of aPKC–Par6 and Lgl activities. Combining structural, biochemical, cellular and in vivo assays, the authors uncover the mechanism for capture and multisite phosphorylation of lethal (2) giant larvae by the atypical protein kinase C and partitioning-defective protein 6, revealing the basis for their mutual antagonism underpinning cell polarity.

aPKC-Par6及其多位点极性底物Lgl的捕获、相互抑制和释放机制
非典型蛋白激酶C (aPKC)和分裂缺陷蛋白6 (Par6)与底物致死巨型幼虫(Lgl)的相互拮抗关系对于调节许多细胞类型的极性至关重要。尽管aPKC-Par6在三个丝氨酸位点磷酸化Lgl以将其排除在顶端结构域之外,但aPKC-Par6和Lgl矛盾地形成了一个稳定的激酶-底物复合物,Par6的作用相互矛盾。我们报道了通过apkci对接位点和Par6PDZ接触点捕获的与全长Llgl1结合的人类apkci - par6α的结构。该复合物捕获aPKC和Par6之间的磷酸化- s663 Llgl1中间桥接,阻碍磷酸化进程。因此,Llgl1pS663有效抑制aPKCι,而Llgl1被aPKCι - par6捕获。Lgl - apkc相互作用的突变破坏阻碍了复合物的组装和Lgl的磷酸化,而破坏Lgl - par6pdz的接触促进了复合物的解离和Lgl的磷酸化。我们展示了par6pdz调节的底物捕获和释放模型,该模型需要活性Cdc42和顶端伙伴碎屑的结合来驱动复杂的分解。我们的研究结果提示了aPKC-Par6和Lgl活性相互调控和空间调控的机制。
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文
来源期刊

Nature Structural & Molecular Biology
BIOCHEMISTRY & MOLECULAR BIOLOGY-BIOPHYSICS
CiteScore
22.00
自引率
1.80%
发文量
160
审稿时长
3-8 weeks
期刊介绍:
Nature Structural & Molecular Biology is a comprehensive platform that combines structural and molecular research. Our journal focuses on exploring the functional and mechanistic aspects of biological processes, emphasizing how molecular components collaborate to achieve a particular function. While structural data can shed light on these insights, our publication does not require them as a prerequisite.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:



