Synthesis and biological evaluation of novel pyrrolo[2,3-b]pyridine derivatives as potent GSK-3β inhibitors for treating Alzheimer's disease
IF 6
2区 医学
Q1 CHEMISTRY, MEDICINAL
引用次数: 0
Abstract
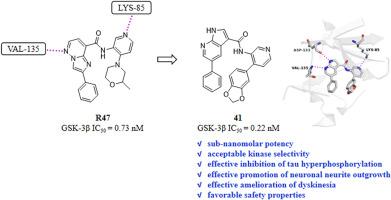
The development of potent glycogen synthase kinase-3β (GSK-3β) inhibitor has been increasingly recognized as the candidate treatment against the multifactorial pathogenic mechanism of Alzheimer's disease (AD). This study prepared various new pyrrolo[2,3-b]pyridine derivatives, evaluated the anti-AD activities and detected the security based on the structure-guided rational design. Our results indicated that many pyrrolo[2,3-b]pyridine derivatives had strong GSK-3β inhibitory activities, particularly compounds 41, 46 and 54, with the half maximal inhibitory concentrations (IC50) of 0.22, 0.26 and 0.24 nM, respectively, and each of them generally possessed GSK-3β selectivity over 24 structurally similar kinases. In addition, further targeting studies at the cellular level revealed that compound 41 increased GSK-3β phosphorylation at Ser9 site dose-dependently for inhibiting GSK-3β activity, therefore inhibiting the hyperphosphorylation of tau protein by decreasing the p-tau-Ser396 abundance. Moreover, 41 up-regulated β-catenin and neurogenesis-related markers (GAP43 and MAP-2), thereby promoting neurite outgrowth of neurons in SH-SY5Y cells. According to the in vitro cells assay, 41 showed the lower cytotoxicity to SH-SY5Y cells with a survival rate of over 70 % at the concentration of 100 μM. In vivo efficacy and acute toxicity experiments showed that, 41 effectively ameliorated the dyskinesia in AlCl3-induced zebrafish AD models and exhibited its low‐toxicity nature in C57BL/6 mice. Overall, the pyrrolo[2,3-b]pyridine derivative 41 could serve as a promising GSK-3β inhibitor for treating AD.

新型吡咯[2,3-b]吡啶衍生物作为抗阿尔茨海默病GSK-3β抑制剂的合成及生物学评价
强效糖原合成酶激酶-3β (GSK-3β)抑制剂的开发越来越被认为是对抗阿尔茨海默病(AD)多因子致病机制的候选治疗方法。本研究制备了多种新型吡咯[2,3-b]吡啶衍生物,并基于结构导向的合理设计对其抗ad活性进行了评价和安全性检测。结果表明,许多吡咯[2,3-b]吡啶衍生物具有较强的GSK-3β抑制活性,特别是化合物41、46和54,其最大半数抑制浓度(IC50)分别为0.22、0.26和0.24 nM,并且它们对24种结构相似的激酶均具有GSK-3β选择性。此外,进一步的细胞水平靶向研究表明,化合物41增加了GSK-3β Ser9位点磷酸化的剂量依赖性,从而抑制GSK-3β活性,从而通过降低p-tau-Ser396丰度来抑制tau蛋白的过度磷酸化。此外,41上调β-catenin和神经发生相关标志物(GAP43和MAP-2),从而促进SH-SY5Y细胞神经元的神经突生长。体外细胞实验显示,在100 μM浓度下,41对SH-SY5Y细胞具有较低的细胞毒性,存活率超过70%。体内药效和急性毒性实验表明,41能有效改善alcl3诱导的斑马鱼AD模型的运动障碍,对C57BL/6小鼠具有低毒性。综上所述,吡咯[2,3-b]吡啶衍生物41可作为治疗AD的GSK-3β抑制剂。
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文
来源期刊
CiteScore
11.70
自引率
9.00%
发文量
863
审稿时长
29 days
期刊介绍:
The European Journal of Medicinal Chemistry is a global journal that publishes studies on all aspects of medicinal chemistry. It provides a medium for publication of original papers and also welcomes critical review papers.
A typical paper would report on the organic synthesis, characterization and pharmacological evaluation of compounds. Other topics of interest are drug design, QSAR, molecular modeling, drug-receptor interactions, molecular aspects of drug metabolism, prodrug synthesis and drug targeting. The journal expects manuscripts to present the rational for a study, provide insight into the design of compounds or understanding of mechanism, or clarify the targets.

 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


