Dysregulation of mTOR signalling is a converging mechanism in lissencephaly
IF 50.5
1区 综合性期刊
Q1 MULTIDISCIPLINARY SCIENCES
引用次数: 0
Abstract
Cerebral cortex development in humans is a highly complex and orchestrated process that is under tight genetic regulation. Rare mutations that alter gene expression or function can disrupt the structure of the cerebral cortex, resulting in a range of neurological conditions1. Lissencephaly (‘smooth brain’) spectrum disorders comprise a group of rare, genetically heterogeneous congenital brain malformations commonly associated with epilepsy and intellectual disability2. However, the molecular mechanisms underlying disease pathogenesis remain unknown. Here we establish hypoactivity of the mTOR pathway as a clinically relevant molecular mechanism in lissencephaly spectrum disorders. We characterized two types of cerebral organoid derived from individuals with genetically distinct lissencephalies with a recessive mutation in p53-induced death domain protein 1 (PIDD1) or a heterozygous chromosome 17p13.3 microdeletion leading to Miller–Dieker lissencephaly syndrome (MDLS). PIDD1-mutant organoids and MDLS organoids recapitulated the thickened cortex typical of human lissencephaly and demonstrated dysregulation of protein translation, metabolism and the mTOR pathway. A brain-selective activator of mTOR complex 1 prevented and reversed cellular and molecular defects in the lissencephaly organoids. Our findings show that a converging molecular mechanism contributes to two genetically distinct lissencephaly spectrum disorders. Cellular, transcriptomic and proteomic analyses of organoids derived from human induced pluripotent stem cells show that mTOR pathway hypoactivation is involved in two genetically distinct lissencephaly spectrum disorders.

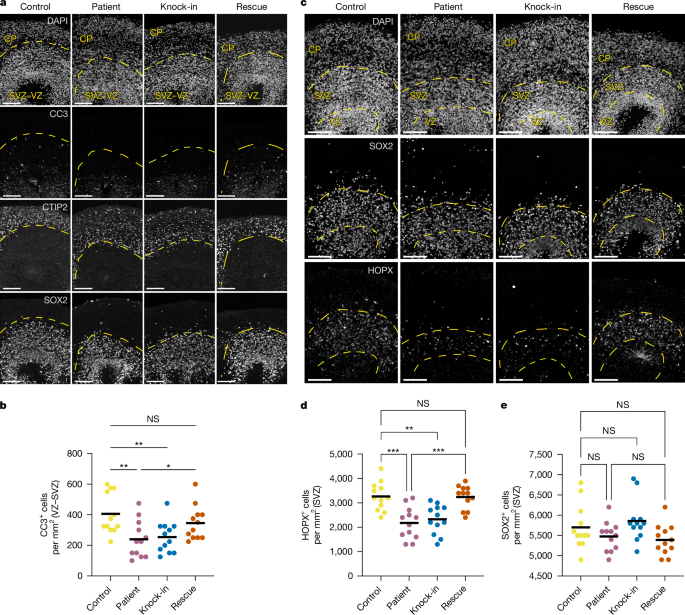
mTOR信号的失调是无脑畸形的一种会聚机制
人类大脑皮层的发育是一个高度复杂和协调的过程,在严格的基因调控下。改变基因表达或功能的罕见突变会破坏大脑皮层的结构,导致一系列神经系统疾病。无脑畸形(“平滑脑”)谱系障碍包括一组罕见的、遗传异质性的先天性脑畸形,通常与癫痫和智力残疾有关2。然而,疾病发病的分子机制尚不清楚。在这里,我们建立mTOR通路的低活性作为无脑谱系障碍的临床相关分子机制。我们鉴定了两种类型的脑类器官,这些脑类器官来自遗传上不同的无脑畸形患者,其中p53诱导的死亡结构域蛋白1 (PIDD1)隐性突变或杂合染色体17p13.3微缺失导致米勒-迪克尔无脑畸形综合征(MDLS)。pidd1突变的类器官和MDLS类器官重现了人类无脑畸形典型的增厚皮层,并表现出蛋白质翻译、代谢和mTOR途径的失调。mTOR复合物1的脑选择性激活剂预防和逆转无脑类器官的细胞和分子缺陷。我们的研究结果表明,趋同的分子机制有助于两种遗传上不同的无脑谱系障碍。
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文
来源期刊

Nature
综合性期刊-综合性期刊
CiteScore
90.00
自引率
1.20%
发文量
3652
审稿时长
3 months
期刊介绍:
Nature is a prestigious international journal that publishes peer-reviewed research in various scientific and technological fields. The selection of articles is based on criteria such as originality, importance, interdisciplinary relevance, timeliness, accessibility, elegance, and surprising conclusions. In addition to showcasing significant scientific advances, Nature delivers rapid, authoritative, insightful news, and interpretation of current and upcoming trends impacting science, scientists, and the broader public. The journal serves a dual purpose: firstly, to promptly share noteworthy scientific advances and foster discussions among scientists, and secondly, to ensure the swift dissemination of scientific results globally, emphasizing their significance for knowledge, culture, and daily life.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


