Multi-omic profiling a defined bacterial consortium for treatment of recurrent Clostridioides difficile infection
IF 58.7
1区 医学
Q1 BIOCHEMISTRY & MOLECULAR BIOLOGY
引用次数: 0
Abstract
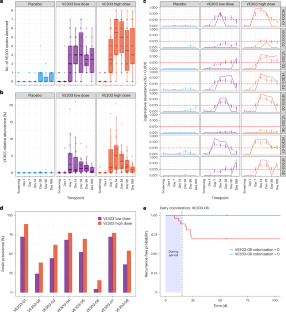
Donor-derived fecal microbiota treatments are efficacious in preventing recurrent Clostridioides difficile infection (rCDI), but they have inherently variable quality attributes, are difficult to scale and harbor the risk of pathogen transfer. In contrast, VE303 is a defined consortium of eight purified, clonal bacterial strains developed for prevention of rCDI. In the phase 2 CONSORTIUM study, high-dose VE303 was well tolerated and reduced the odds of rCDI by more than 80% compared to placebo. VE303 organisms robustly colonized the gut in the high-dose group and were among the top taxa associated with non-recurrence. Multi-omic modeling identified antibiotic history, baseline stool metabolites and serum cytokines as predictors of both on-study CDI recurrence and VE303 colonization. VE303 potentiated early recovery of the host microbiome and metabolites with increases in short-chain fatty acids, secondary bile acids and bile salt hydrolase genes after antibiotic treatment for CDI, which is considered important to prevent CDI recurrences. These results support the idea that VE303 promotes efficacy in rCDI through multiple mechanisms. Results of multi-omic profiling of the microbiome and host immunity of individuals treated with VE303 to prevent recurrent Clostridioides difficile infection in the context of a phase 2 trial show robust colonization of VE303 and indicate potential biomarkers of response.

多组学分析确定的细菌联合体治疗复发性难辨梭状芽胞杆菌感染
供体来源的粪便微菌群治疗在预防复发性艰难梭菌感染(rCDI)方面是有效的,但它们具有固有的可变质量属性,难以规模化,并且存在病原体转移的风险。相比之下,VE303是一个由8个纯化的克隆菌株组成的确定的联盟,用于预防rCDI。在2期联合研究中,与安慰剂相比,高剂量VE303耐受性良好,将rCDI的几率降低了80%以上。在高剂量组中,VE303微生物在肠道中稳定定植,并且是与不复发相关的顶级分类群之一。多组学模型确定抗生素史、基线粪便代谢物和血清细胞因子是研究中CDI复发和VE303定植的预测因子。抗生素治疗CDI后,VE303通过增加短链脂肪酸、次级胆汁酸和胆盐水解酶基因,增强宿主微生物组和代谢物的早期恢复,这被认为是预防CDI复发的重要因素。这些结果支持了VE303通过多种机制促进rCDI疗效的观点。
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文
来源期刊

Nature Medicine
医学-生化与分子生物学
CiteScore
100.90
自引率
0.70%
发文量
525
审稿时长
1 months
期刊介绍:
Nature Medicine is a monthly journal publishing original peer-reviewed research in all areas of medicine. The publication focuses on originality, timeliness, interdisciplinary interest, and the impact on improving human health. In addition to research articles, Nature Medicine also publishes commissioned content such as News, Reviews, and Perspectives. This content aims to provide context for the latest advances in translational and clinical research, reaching a wide audience of M.D. and Ph.D. readers. All editorial decisions for the journal are made by a team of full-time professional editors.
Nature Medicine consider all types of clinical research, including:
-Case-reports and small case series
-Clinical trials, whether phase 1, 2, 3 or 4
-Observational studies
-Meta-analyses
-Biomarker studies
-Public and global health studies
Nature Medicine is also committed to facilitating communication between translational and clinical researchers. As such, we consider “hybrid” studies with preclinical and translational findings reported alongside data from clinical studies.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


