The blue-light receptor CRY1 serves as a switch to balance photosynthesis and plant defense
IF 20.6
1区 医学
Q1 MICROBIOLOGY
引用次数: 0
Abstract
Plant stomata open in response to blue light, allowing gas exchange and water transpiration. However, open stomata are potential entry points for pathogens. Whether plants can sense pathogens and mount defense responses upon stomatal opening and how blue-light cues are integrated to balance growth-defense trade-offs are poorly characterized. We show that the Arabidopsis blue-light photoreceptor CRYPTOCHROME 1 (CRY1) mediates various aspects of immunity, including pathogen-triggered stomatal closure as well as activation of plant immunity through a typical light-responsive protein LATE UPREGULATED IN RESPONSE TO HYALOPERONOSPORA PARASITICA (LURP1). LURP1 undergoes N-terminal palmitoylation in the presence of bacterial flagellin, prompting a change in subcellular localization from the cytoplasm to plasma membrane, where it enhances the activity of the receptor FLAGELLIN SENSING 2 (FLS2) to mediate plant defense. Collectively, these findings reveal that blue light regulates stomatal defense and highlight the dual functions of CRY1 in photosynthesis and immunity.
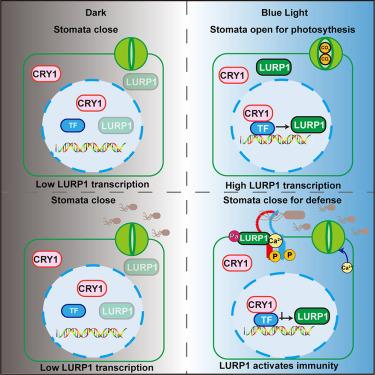
蓝光受体CRY1是平衡光合作用和植物防御的开关
植物的气孔在蓝光下打开,允许气体交换和水分蒸腾。然而,开放的气孔是病原体的潜在入口。植物是否可以感知病原体并在气孔打开时进行防御反应,以及蓝光信号如何被整合以平衡生长-防御权衡,这些都没有得到很好的描述。我们发现拟南芥蓝光光感受器CRYPTOCHROME 1 (CRY1)介导免疫的各个方面,包括病原体触发的气孔关闭,以及通过一个典型的光响应蛋白对HYALOPERONOSPORA PARASITICA (LURP1)的晚上调来激活植物免疫。在细菌鞭毛蛋白存在的情况下,LURP1发生n端棕榈酰化,促使亚细胞定位从细胞质到质膜的变化,从而增强鞭毛蛋白受体FLS2的活性,介导植物防御。综上所述,这些发现揭示了蓝光调节气孔防御,并突出了CRY1在光合作用和免疫中的双重功能。
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文
来源期刊

Cell host & microbe
生物-微生物学
CiteScore
45.10
自引率
1.70%
发文量
201
审稿时长
4-8 weeks
期刊介绍:
Cell Host & Microbe is a scientific journal that was launched in March 2007. The journal aims to provide a platform for scientists to exchange ideas and concepts related to the study of microbes and their interaction with host organisms at a molecular, cellular, and immune level. It publishes novel findings on a wide range of microorganisms including bacteria, fungi, parasites, and viruses. The journal focuses on the interface between the microbe and its host, whether the host is a vertebrate, invertebrate, or plant, and whether the microbe is pathogenic, non-pathogenic, or commensal. The integrated study of microbes and their interactions with each other, their host, and the cellular environment they inhabit is a unifying theme of the journal. The published work in Cell Host & Microbe is expected to be of exceptional significance within its field and also of interest to researchers in other areas. In addition to primary research articles, the journal features expert analysis, commentary, and reviews on current topics of interest in the field.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


