Unravelling the reaction mechanism on Ni–YSZ anode supported direct ammonia solid fuel cell: Experimental and theoretical studies
IF 6.3
2区 材料科学
Q2 CHEMISTRY, PHYSICAL
引用次数: 0
Abstract
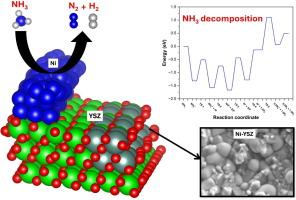
While the state-of-the-art nickel–yttria-stabilized zirconia (Ni–YSZ) cermet exhibits a great potential to realize high-efficiency ammonia to power conversion using direct ammonia solid oxide fuel cell (DA-SOFC), the mechanisms of NH3 cracking and H2 oxidation reaction are still unclear. In this context, with the objective of enhancing Ni–YSZ anode activity, the performance of DA-SOFC using conventional (Ni–YSZ|YSZ|GDC–LSCF) anode-supported cell is studied through in-depth materials characterisation coupled with density functional theory (DFT) calculations to unveil the reaction’s mechanism on Ni–YSZ(111) anode. The performance of SOFC is examined under the NH3, H2, and N2/H2 mixtures as fuel at three different temperatures: 700, 750, and 800 °C. In addition to X-ray diffraction (XRD) and scanning electron microscopy coupled with energy dispersive X-ray spectroscopy (SEM–EDX) characterizations, the electrochemical characterization of Ni–YSZ anode is performed using the electochemical impedance spectroscopy (EIS) technique at 700–800 °C. Notably, polarization resistance is primarily due to the charge transfer reaction and secondarily by the gas diffusion impedance observed. The activation barriers of elementary steps involved in the proposed mechanisms for NH3 cracking and H2 oxidation reactions on the Ni–YSZ(111) surface are computed using ab initio calculations. DFT findings revealed that the hydrogenation step of hydroxyl species, accompanied by charge transfer, dominates the hydrogen oxidation reaction. In contrast, the recombination and desorption of nitrogen adatoms act as the rate-controlling step for NH3 decomposition, demonstrating the highest kinetic barrier of all elementary steps. This study offers a mechanistic understanding that is vital for achieving the rational design of Ni–YSZ-based anode cermet for DA-SOFC.

Ni-YSZ阳极负载的直接氨固体燃料电池反应机理的实验与理论研究
虽然最先进的镍钇稳定氧化锆(Ni-YSZ)金属陶瓷在直接氨固体氧化物燃料电池(DA-SOFC)中显示出实现高效氨制电的巨大潜力,但NH3裂解和H2氧化反应的机理尚不清楚。在此背景下,为了提高Ni-YSZ(111)阳极的活性,通过深入的材料表征和密度泛函理论(DFT)计算,研究了传统NiO-Y2O3-ZrO2| ce0.8 gd0.2 2o1.95 - la0.6 sr0.4 co0.2 fe0.8 o3 NiO-Y2O3-ZrO2| ce0.8 gd0.2 2o1.95 - la0.6 sr0.4 co0.2 fe0.8 o3 (Ni-YSZ |YSZ| GDC-LSCF)阳极负载电池的DA-SOFC性能,揭示了Ni-YSZ(111)阳极上的反应机理。在700、750和800 °C三种不同温度下,以NH3、H2和N2/H2混合物作为燃料测试SOFC的性能。除了x射线衍射(XRD)和扫描电镜结合能量色散x射线能谱(SEM-EDX)表征外,还使用电化学阻抗谱(EIS)技术在700-800 °C下对Ni-YSZ阳极进行了电化学表征。值得注意的是,极化电阻主要由电荷转移反应引起,其次由观察到的气体扩散阻抗引起。采用从头算方法计算了Ni-YSZ(111)表面NH3裂解和H2氧化反应机制中涉及的基本步骤的激活势垒。DFT结果表明,氢氧化反应以羟基加氢步骤为主,并伴有电荷转移。相反,氮原子的重组和解吸是NH3分解的速率控制步骤,表现出所有基本步骤中最高的动力学势垒。该研究为实现合理设计ni - ysz基DA-SOFC阳极陶瓷提供了重要的机理理解。
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文
来源期刊

Applied Surface Science
工程技术-材料科学:膜
CiteScore
12.50
自引率
7.50%
发文量
3393
审稿时长
67 days
期刊介绍:
Applied Surface Science covers topics contributing to a better understanding of surfaces, interfaces, nanostructures and their applications. The journal is concerned with scientific research on the atomic and molecular level of material properties determined with specific surface analytical techniques and/or computational methods, as well as the processing of such structures.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


