Impurity Control Strategy of TMS-Triol in the Manufacture Development of Islatravir
IF 3.1
3区 化学
Q2 CHEMISTRY, APPLIED
引用次数: 0
Abstract
The identification, quantification, understanding, and control of impurities in starting materials, intermediates, and substances are crucial during the evaluation of new molecular entities in the clinical development of the pharmaceutical industry. Through process characterization development in islatravir (MK-8591), two impurities from the regulatory starting material, TMS-Triol, were discovered in a late stage that persisted in the product. However, they were not detectable by the gas chromatography (GC) method that the process used. This paper describes the use of orthogonal analytical approaches for impurity investigations, which aim to provide a comprehensive understanding of the impurity profile of a drug substance with a particular emphasis on comprehending the origin and fate of impurities from upstream steps. The understanding serves as a guide for making decisions on process controls and optimizations of the islatravir drug substance. This paper also outlines quality control strategies for the starting material of the islatravir drug substance commercial process. The knowledge gained from these investigations forms a solid foundation for establishing specifications for impurities in regulatory starting materials.
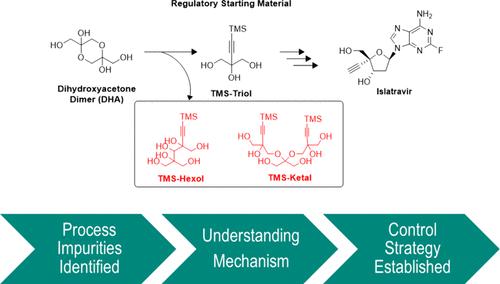
伊斯拉他韦生产开发中tms -三醇的杂质控制策略
在制药工业的临床开发中,对起始原料、中间体和物质中杂质的鉴定、定量、理解和控制是评价新分子实体的关键。通过对islatravir (MK-8591)的工艺表征开发,在后期发现了两种来自监管起始材料TMS-Triol的杂质,这些杂质持续存在于产品中。然而,该工艺使用的气相色谱(GC)方法无法检测到它们。本文描述了使用正交分析方法进行杂质调查,其目的是提供对原料药杂质概况的全面了解,特别强调了解上游步骤中杂质的来源和命运。该理解可作为对依拉他韦原料药的工艺控制和优化决策的指导。本文还概述了依拉他韦原料药商业化过程中起始原料的质量控制策略。从这些调查中获得的知识为建立法规起始材料中杂质的规范奠定了坚实的基础。
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文
来源期刊
CiteScore
6.90
自引率
14.70%
发文量
251
审稿时长
2 months
期刊介绍:
The journal Organic Process Research & Development serves as a communication tool between industrial chemists and chemists working in universities and research institutes. As such, it reports original work from the broad field of industrial process chemistry but also presents academic results that are relevant, or potentially relevant, to industrial applications. Process chemistry is the science that enables the safe, environmentally benign and ultimately economical manufacturing of organic compounds that are required in larger amounts to help address the needs of society. Consequently, the Journal encompasses every aspect of organic chemistry, including all aspects of catalysis, synthetic methodology development and synthetic strategy exploration, but also includes aspects from analytical and solid-state chemistry and chemical engineering, such as work-up tools,process safety, or flow-chemistry. The goal of development and optimization of chemical reactions and processes is their transfer to a larger scale; original work describing such studies and the actual implementation on scale is highly relevant to the journal. However, studies on new developments from either industry, research institutes or academia that have not yet been demonstrated on scale, but where an industrial utility can be expected and where the study has addressed important prerequisites for a scale-up and has given confidence into the reliability and practicality of the chemistry, also serve the mission of OPR&D as a communication tool between the different contributors to the field.

 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


