Dehydrogenative Coupling of Alcohols with Hydrazines under Nickel Catalysis
IF 3.3
2区 化学
Q1 CHEMISTRY, ORGANIC
引用次数: 0
Abstract
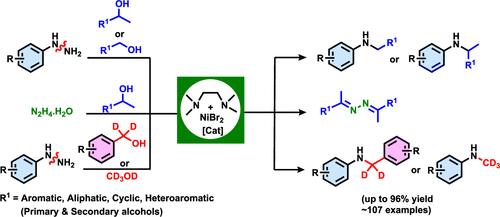
The development of efficient and robust catalytic systems based on earth-abundant transition metals for fundamentally new transformations is crucial for sustainable chemical synthesis. Herein, an effective and selective Ni-catalyzed dehydrogenative coupling of alcohols with hydrazines with the liberation of ammonia gas is reported. Although several methods were documented for the N-alkylation reaction, the present strategy is conceptually novel, and the reaction proceeds through a pathway involving N–N bond cleavage of phenylhydrazine followed by hydrogen autotransfer. This unprecedented method demonstrates a wide substrate scope, allowing for the synthesis of C–N coupled products from arylhydrazines using various types of alcohols, including aryl, fused aryl, heteroaromatic, cyclic, and aliphatic alcohols, both primary and secondary alcohols. The present catalytic approach was expanded to facilitate selective deuterium incorporation reactions by employing deuterated alcohols at the α-methyl position of the resulting N-alkylated products. It is noteworthy that we have broadened the applicability of the current catalytic systems to facilitate the ketazine synthesis of hydrazine monohydrate by employing secondary alcohols. The reaction utilizes an inexpensive, abundant, and renewable alcohol that serves as both an alkylating and (transfer) hydrogenating agent. Kinetic studies reveal that the reaction rate depends on the concentration of arylhydrazine and the nickel catalyst, following fractional order.
求助全文
约1分钟内获得全文
求助全文
来源期刊

Journal of Organic Chemistry
化学-有机化学
CiteScore
6.20
自引率
11.10%
发文量
1467
审稿时长
2 months
期刊介绍:
Journal of Organic Chemistry welcomes original contributions of fundamental research in all branches of the theory and practice of organic chemistry. In selecting manuscripts for publication, the editors place emphasis on the quality and novelty of the work, as well as the breadth of interest to the organic chemistry community.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


