Enhanced glucose-to-5-hydroxymethylfurfural transformation activity over CePO4 catalyst: Insights into crystal structure, acidic property and reaction pathway
IF 6.5
1区 化学
Q2 CHEMISTRY, PHYSICAL
引用次数: 0
Abstract
The conversion of carbohydrates to high value-added 5-hydroxymethylfurfural (5-HMF) has aroused intensive research interest. Herein, a series of cerium phosphates was prepared by simple calcination accompanied by a hexagonal-monoclinic phase transformation. Hexagonal CePO4-200 exhibits superior catalytic activity for the conversion of glucose to 5-HMF with a yield of 73 % compared with monoclinic CePO4-800. The related characterization results confirm that hexagonal CePO4-200 possesses unoccupied and open spaces leading to favorable coordination environments, good pore structure and high acid density, which contributed to activation and catalysis of glucose. Density functional theory (DFT) calculations proves that the hexagonal CePO4-200 interface possesses a more effective charge transfer ability by comparing with monoclinic CePO4-800 interface in Lewis acidic Ce3+ sites region thus enhancing the catalytic activity. Additionally, a feasible tandem reaction mechanism through glucose-to-fructose isomerization driven by Ce3+ acidic sites and fructose dehydration to 5-HMF catalyzed by P-OH group was proposed.
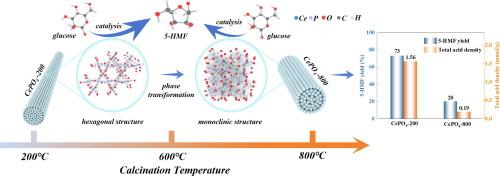

增强 CePO4 催化剂的葡萄糖-5-羟甲基糠醛转化活性:晶体结构、酸性和反应途径的启示
碳水化合物转化为高附加值的5-羟甲基糠醛(5-HMF)引起了广泛的研究兴趣。本文采用简单煅烧的方法制备了一系列磷酸铈,并进行了六方-单斜相变。与单斜CePO4-800相比,六方CePO4-200在葡萄糖转化为5-羟甲基糠醛方面表现出更好的催化活性,产率为73 %。相关表征结果证实,六方CePO4-200具有空位和开放空间,具有良好的配位环境、良好的孔隙结构和较高的酸密度,有助于葡萄糖的活化和催化。密度泛函理论(DFT)计算证明,在Lewis酸性Ce3+位区,六方CePO4-200界面比单斜CePO4-800界面具有更有效的电荷转移能力,从而提高了催化活性。此外,提出了一种可行的串联反应机制,即Ce3+酸性位点驱动葡萄糖-果糖异构化,P-OH基团催化果糖脱水生成5-HMF。
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文
来源期刊

Journal of Catalysis
工程技术-工程:化工
CiteScore
12.30
自引率
5.50%
发文量
447
审稿时长
31 days
期刊介绍:
The Journal of Catalysis publishes scholarly articles on both heterogeneous and homogeneous catalysis, covering a wide range of chemical transformations. These include various types of catalysis, such as those mediated by photons, plasmons, and electrons. The focus of the studies is to understand the relationship between catalytic function and the underlying chemical properties of surfaces and metal complexes.
The articles in the journal offer innovative concepts and explore the synthesis and kinetics of inorganic solids and homogeneous complexes. Furthermore, they discuss spectroscopic techniques for characterizing catalysts, investigate the interaction of probes and reacting species with catalysts, and employ theoretical methods.
The research presented in the journal should have direct relevance to the field of catalytic processes, addressing either fundamental aspects or applications of catalysis.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


