Identification of Novel Human 15-Lipoxygenase-2 (h15-LOX-2) Inhibitors Using a Virtual Screening Approach
IF 6.8
1区 医学
Q1 CHEMISTRY, MEDICINAL
引用次数: 0
Abstract
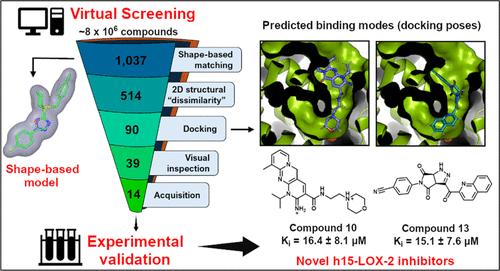
The human 15-lipoxygenase-2 (h15-LOX-2) catalyzes mainly the regio- and stereospecific oxygenation of arachidonate to its corresponding hydroperoxide (15(S)-HpETE). h15-LOX-2 is implicated in the biosynthesis of inflammatory lipid mediators and plays a role in the development of atherosclerotic plaques, but it is still underexploited as a drug target. Here, to search for novel h15-LOX-2 inhibitors, we used a virtual screening (VS) approach consisting of shape-based matching, two-dimensional (2D) structural “dissimilarity”, docking, and visual inspection filters, which were applied to a “curated” ZINC database (∼8 × 106 compounds). The VS was experimentally validated, and six micromolar-range inhibitors were identified among 13 tested compounds (46.2%). The Ki values could be determined for two inhibitors, compounds 10 (Ki = 16.4 ± 8.1 μM) and 13 (Ki = 15.1 ± 7.6 μM), which showed a mixed-type mechanism of inhibition. Overall, the identified inhibitors fulfill drug-like criteria and are structurally novel compared with known h15-LOX-2 inhibitors.
利用虚拟筛选方法鉴定新型人15-脂氧合酶-2 (h15-LOX-2)抑制剂
人15-脂氧合酶-2 (h15-LOX-2)主要催化花生四烯酸酯的区域和立体特异性氧合成相应的过氧化氢(15(S)- hpete)。h15-LOX-2参与炎症脂质介质的生物合成,并在动脉粥样硬化斑块的形成中发挥作用,但作为药物靶点仍未得到充分开发。在这里,为了寻找新的h15-LOX-2抑制剂,我们使用了一种虚拟筛选(VS)方法,包括基于形状的匹配、二维(2D)结构“不相似性”、对接和视觉检查过滤器,这些方法应用于“精心设计的”锌数据库(约8 × 106种化合物)。实验验证了该方法的有效性,在13个被测化合物中鉴定出6个微摩尔范围的抑制剂(46.2%)。化合物10 (Ki = 16.4±8.1 μM)和化合物13 (Ki = 15.1±7.6 μM)的Ki值均可测定,表现出混合型抑制机制。总的来说,确定的抑制剂满足药物样标准,与已知的h15-LOX-2抑制剂相比,结构新颖。
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文
来源期刊

Journal of Medicinal Chemistry
医学-医药化学
CiteScore
4.00
自引率
11.00%
发文量
804
审稿时长
1.9 months
期刊介绍:
The Journal of Medicinal Chemistry is a prestigious biweekly peer-reviewed publication that focuses on the multifaceted field of medicinal chemistry. Since its inception in 1959 as the Journal of Medicinal and Pharmaceutical Chemistry, it has evolved to become a cornerstone in the dissemination of research findings related to the design, synthesis, and development of therapeutic agents.
The Journal of Medicinal Chemistry is recognized for its significant impact in the scientific community, as evidenced by its 2022 impact factor of 7.3. This metric reflects the journal's influence and the importance of its content in shaping the future of drug discovery and development. The journal serves as a vital resource for chemists, pharmacologists, and other researchers interested in the molecular mechanisms of drug action and the optimization of therapeutic compounds.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


