Non-tandem enzymatic modules-encapsulated metal-organic framework cubes with mutual-validated operation for dual-modal competitive immunoassay of antibiotics
IF 8
1区 化学
Q1 CHEMISTRY, ANALYTICAL
引用次数: 0
Abstract
Adaptive fusion of nanoscale metal-organic frameworks (nMOFs) and enzyme-based immunoassay is an insightful combination for inspired establishment of antibiotic assay, typically in a competitive format, in which inherent enzymes in nMOFs can serve as generators for amplifying catalytic signals. Albeit important, there is currently a lack of explorations of enzymatic signal outputs with multiple modalities in typical competitive immunoassays, which may face the false signals and lack cross-validation. Herein, non-tandem bi-enzymatic modules such as urease and horseradish peroxidase were co-encapsulated into nMOF cubes and functionalized with antibodies into entities for dual-modal competitive immunoassay of oxytetracycline (OTC), which can convert the bi-enzymatic catalytic signals into fluorogenic and chromogenic reflection and can gain the mutual validation merits. The fluorescence mode can achieve an OTC detection range from 5 ng/mL to 2000 ng/mL, with a detection limit (LOD) of 2.14 ng/mL; complementally, the absorbance mode can achieve a relatively narrow detection range from 1 ng/mL to 200 ng/mL, but a lower LOD of 0.32 ng/mL. The dual-mode sensing parameters of the competitive immunoassay in responding to OTC analysis differed in terms of the linear range and detection limit, which can be integrated to offer reciprocal authentication and expand the detection diversity. Overall, our proposed method may provide a new idea in dual-modal competitive immunoassay of antibiotics, signifying its promising expansion for rational substitution of enzymatic modules and its potential performance in wide applications.
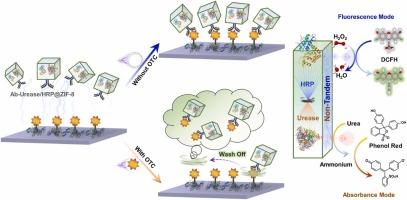
非串联酶模块-封装金属-有机框架立方体与相互验证操作的双模态竞争性抗生素免疫分析
纳米级金属有机框架(nMOFs)与基于酶的免疫测定的自适应融合是建立抗生素检测的一个有见地的组合,通常采用竞争形式,其中 nMOFs 中固有的酶可作为放大催化信号的发生器。尽管这很重要,但目前缺乏对典型竞争性免疫测定中多种模式的酶信号输出的探索,这可能会出现假信号并缺乏交叉验证。本文将脲酶和辣根过氧化物酶等非串联双酶模块共同封装到nMOF立方体中,并将抗体功能化为土霉素(OTC)双模态竞争性免疫测定的实体,可将双酶催化信号转化为荧光和色原反映,获得相互验证的优点。荧光模式可实现从 5 纳克/毫升到 2000 纳克/毫升的 OTC 检测范围,检出限(LOD)为 2.14 纳克/毫升;作为补充,吸光模式可实现从 1 纳克/毫升到 200 纳克/毫升的相对较窄的检测范围,但检出限(LOD)较低,为 0.32 纳克/毫升。竞争性免疫测定的双模式传感参数在响应 OTC 分析时的线性范围和检出限方面存在差异,可将其整合以提供相互认证并扩大检测多样性。总之,我们提出的方法为抗生素的双模式竞争性免疫分析提供了一种新思路,标志着其在合理替代酶模块方面具有广阔的发展前景和广泛的应用潜力。
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文
来源期刊

Sensors and Actuators B: Chemical
工程技术-电化学
CiteScore
14.60
自引率
11.90%
发文量
1776
审稿时长
3.2 months
期刊介绍:
Sensors & Actuators, B: Chemical is an international journal focused on the research and development of chemical transducers. It covers chemical sensors and biosensors, chemical actuators, and analytical microsystems. The journal is interdisciplinary, aiming to publish original works showcasing substantial advancements beyond the current state of the art in these fields, with practical applicability to solving meaningful analytical problems. Review articles are accepted by invitation from an Editor of the journal.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


