Superatomic Stabilization of Dinuclear Platinum(III) through Iodide-Bridged Five-Center Ten-Electron Bonding
IF 4.7
2区 化学
Q1 CHEMISTRY, INORGANIC & NUCLEAR
引用次数: 0
Abstract
One of the goals in synthetic chemistry is to obtain compounds featuring unusual valence states that are stable under ambient conditions. At present, stabilizing unusual Pt(III) states is considered difficult, except through direct Pt–Pt bonding such as that in platinum-blues or organometallization using bulky ligands. Pt(III) stabilization is also very difficult in halogen-bridged metal complex chains (MX-Chains). Herein, the iodide-bridged Pt(III) dimer compound [Pt2(en)4I3]I3 (en = ethylenediamine), which is prepared by the iodine oxidation of [PtII(en)2]I2, has been successfully synthesized and characterized. This compound is stable and is obtained as diamond-shaped single crystals with a lustrous emerald-green color under reflected light and a red color under transmitted light. The Pt(III) state is stabilized by the five-center ten-electron (5c-10e) bonding in the I–Pt–I–Pt–I core, in addition to the very strong antiferromagnetic state. The stabilization mechanism of Pt(III) through a 5c-10e bonding is considered a superatom complex; thus, this work provides new insight for stabilizing the unusual Pt(III) state.
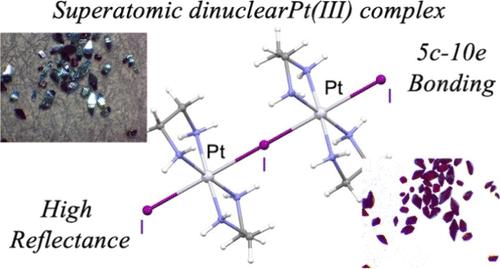
碘化物桥接五中心十电子键对双核铂(III)超原子稳定性的影响
合成化学的目标之一是获得具有在环境条件下稳定的不寻常价态的化合物。目前,稳定不寻常的Pt(III)状态被认为是困难的,除非通过直接的Pt - Pt键合,如铂蓝或使用大块配体的有机金属化。铂(III)在卤素桥接金属络合物链(MX-Chains)中也很难稳定。本文成功合成了由[PtII(en)2]I2经碘氧化法制备的碘桥化Pt(III)二聚体化合物[Pt2(en)4I3]I3 (en =乙二胺)。这种化合物是稳定的,在反射光下呈有光泽的祖母绿,在透射光下呈红色的钻石状单晶。Pt(III)态除了具有很强的反铁磁态外,还被I-Pt-I-Pt-I核中的五中心十电子键(5c-10e)所稳定。Pt(III)通过5c-10e键的稳定机制被认为是一个超原子配合物;因此,这项工作为稳定不寻常的Pt(III)状态提供了新的见解。
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文
来源期刊

Inorganic Chemistry
化学-无机化学与核化学
CiteScore
7.60
自引率
13.00%
发文量
1960
审稿时长
1.9 months
期刊介绍:
Inorganic Chemistry publishes fundamental studies in all phases of inorganic chemistry. Coverage includes experimental and theoretical reports on quantitative studies of structure and thermodynamics, kinetics, mechanisms of inorganic reactions, bioinorganic chemistry, and relevant aspects of organometallic chemistry, solid-state phenomena, and chemical bonding theory. Emphasis is placed on the synthesis, structure, thermodynamics, reactivity, spectroscopy, and bonding properties of significant new and known compounds.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


