Inflammation switches the chemoattractant requirements for naive lymphocyte entry into lymph nodes
IF 45.5
1区 生物学
Q1 BIOCHEMISTRY & MOLECULAR BIOLOGY
引用次数: 0
Abstract
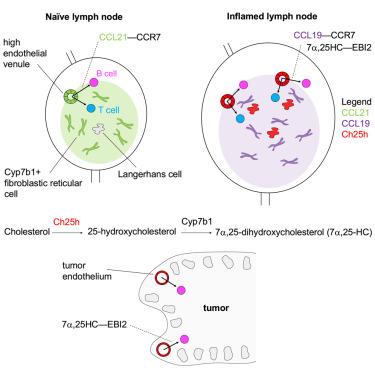
Sustained lymphocyte migration from blood into lymph nodes (LNs) is important for immune responses. The CC-chemokine receptor-7 (CCR7) ligand CCL21 is required for LN entry but is downregulated during inflammation, and it has been unclear how recruitment is maintained. Here, we show that the oxysterol biosynthetic enzyme cholesterol-25-hydroxylase (Ch25h) is upregulated in LN high endothelial venules during viral infection. Lymphocytes become dependent on oxysterols, generated through a transcellular endothelial-fibroblast metabolic pathway, and the receptor EBI2 for inflamed LN entry. Additionally, Langerhans cells are an oxysterol source. Ch25h is also expressed in inflamed peripheral endothelium, and EBI2 mediates B cell recruitment in a tumor model. Finally, we demonstrate that LN CCL19 is critical in lymphocyte recruitment during inflammation. Thus, our work explains how naive precursor trafficking is sustained in responding LNs, identifies a role for oxysterols in cell recruitment into inflamed tissues, and establishes a logic for the CCR7 two-ligand system.
求助全文
约1分钟内获得全文
求助全文
来源期刊
Cell
生物-生化与分子生物学
CiteScore
110.00
自引率
0.80%
发文量
396
审稿时长
2 months
期刊介绍:
Cells is an international, peer-reviewed, open access journal that focuses on cell biology, molecular biology, and biophysics. It is affiliated with several societies, including the Spanish Society for Biochemistry and Molecular Biology (SEBBM), Nordic Autophagy Society (NAS), Spanish Society of Hematology and Hemotherapy (SEHH), and Society for Regenerative Medicine (Russian Federation) (RPO).
The journal publishes research findings of significant importance in various areas of experimental biology, such as cell biology, molecular biology, neuroscience, immunology, virology, microbiology, cancer, human genetics, systems biology, signaling, and disease mechanisms and therapeutics. The primary criterion for considering papers is whether the results contribute to significant conceptual advances or raise thought-provoking questions and hypotheses related to interesting and important biological inquiries.
In addition to primary research articles presented in four formats, Cells also features review and opinion articles in its "leading edge" section, discussing recent research advancements and topics of interest to its wide readership.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


