A gut commensal protozoan determines respiratory disease outcomes by shaping pulmonary immunity
IF 45.5
1区 生物学
Q1 BIOCHEMISTRY & MOLECULAR BIOLOGY
引用次数: 0
Abstract
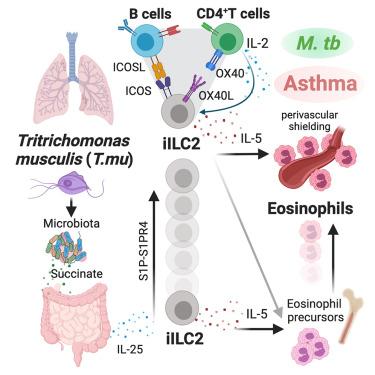
The underlying mechanisms used by the intestinal microbiota to shape disease outcomes of the host are poorly understood. Here, we show that the gut commensal protozoan, Tritrichomonas musculis (T.mu), remotely shapes the lung immune landscape to facilitate perivascular shielding of the airways by eosinophils. Lung-specific eosinophilia requires a tripartite immune network between gut-derived inflammatory group 2 innate lymphoid cells and lung-resident T cells and B cells. This network exacerbates the severity of allergic airway inflammation while hindering the systemic dissemination of pulmonary Mycobacterium tuberculosis. The identification of protozoan DNA sequences in the sputum of patients with severe allergic asthma further emphasizes the relevance of commensal protozoa in human disease. Collectively, these findings demonstrate that a commensal protozoan tunes pulmonary immunity via a gut-operated lung immune network, promoting both beneficial and detrimental disease outcomes in response to environmental airway allergens and pulmonary infections.
求助全文
约1分钟内获得全文
求助全文
来源期刊
Cell
生物-生化与分子生物学
CiteScore
110.00
自引率
0.80%
发文量
396
审稿时长
2 months
期刊介绍:
Cells is an international, peer-reviewed, open access journal that focuses on cell biology, molecular biology, and biophysics. It is affiliated with several societies, including the Spanish Society for Biochemistry and Molecular Biology (SEBBM), Nordic Autophagy Society (NAS), Spanish Society of Hematology and Hemotherapy (SEHH), and Society for Regenerative Medicine (Russian Federation) (RPO).
The journal publishes research findings of significant importance in various areas of experimental biology, such as cell biology, molecular biology, neuroscience, immunology, virology, microbiology, cancer, human genetics, systems biology, signaling, and disease mechanisms and therapeutics. The primary criterion for considering papers is whether the results contribute to significant conceptual advances or raise thought-provoking questions and hypotheses related to interesting and important biological inquiries.
In addition to primary research articles presented in four formats, Cells also features review and opinion articles in its "leading edge" section, discussing recent research advancements and topics of interest to its wide readership.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


