Colorimetric response and adsorption performance of functional granular polyacrylonitrile for Au(I) in thiosulfate solution
IF 8.1
1区 工程技术
Q1 ENGINEERING, CHEMICAL
引用次数: 0
Abstract
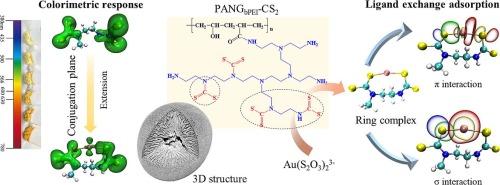
The thiosulfate gold extraction technology represents a typical non-cyanide method, which poses challenges in achieving high efficiency and cost-effective gold recovery. The present study involved the synthesis of a colorimetrically responsive granular polyacrylonitrile material (PANGbPEI-CS2) capable of effectively adsorbing Au(I) in thiosulfate solution. The results show that PANGbPEI-CS2 exhibits a colorimetric response to Au(I) in thiosulfate solution, with the observed visual color transitioning from pale yellow hue to orange. This phenomenon can be attributed to the conjugation plane extension on PANGbPEI-CS2 during Au(I) adsorption, resulting in a redshift of the absorption spectrum towards the visible region. The adsorption of gold in thiosulfate solution by PANGbPEI-CS2 follows the Langmuir isotherm and pseudo-second-order kinetic models, with the maximum loading capacity of 60.502 mg·g−1. The gold adsorption mechanism by PANGbPEI-CS2 involves ligand exchange, wherein gold forms a ring complex with the proximity or adjacency of S atoms on PANGbPEI-CS2. And the adsorption process is influenced by both the pH of the solution and the concentration of thiosulfate. The loaded gold on PANGbPEI-CS2 can be effectively desorbed with acid thiourea solution. This finding are instrumental in addressing the challenges associated with the recovery and rapid detection of gold in thiosulfate solution.

功能性颗粒聚丙烯腈对硫代硫酸溶液中金(I)的比色反应和吸附性能
硫代硫酸盐提金技术是一种典型的非氰化提金方法,对实现高效、低成本的金回收提出了挑战。本研究涉及合成一种比色响应的颗粒状聚丙烯腈材料(PANGbPEI-CS2),该材料能够有效吸附硫代硫酸盐溶液中的Au(I)。结果表明,PANGbPEI-CS2在硫代硫酸盐溶液中对Au(I)表现出比色响应,视觉颜色由淡黄色过渡到橙色。这一现象可归因于在Au(I)吸附过程中,PANGbPEI-CS2上的共轭面扩展,导致吸收光谱向可见光区红移。PANGbPEI-CS2对硫代硫酸盐溶液中金的吸附符合Langmuir等温线和拟二级动力学模型,最大吸附量为60.502 mg·g−1。PANGbPEI-CS2吸附金的机制涉及配体交换,金在PANGbPEI-CS2上与S原子接近或邻接形成环状配合物。吸附过程受溶液pH和硫代硫酸盐浓度的影响。硫脲酸溶液可以有效解吸负载在PANGbPEI-CS2上的金。这一发现有助于解决与硫代硫酸盐溶液中金的回收和快速检测相关的挑战。
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文
来源期刊

Separation and Purification Technology
工程技术-工程:化工
CiteScore
14.00
自引率
12.80%
发文量
2347
审稿时长
43 days
期刊介绍:
Separation and Purification Technology is a premier journal committed to sharing innovative methods for separation and purification in chemical and environmental engineering, encompassing both homogeneous solutions and heterogeneous mixtures. Our scope includes the separation and/or purification of liquids, vapors, and gases, as well as carbon capture and separation techniques. However, it's important to note that methods solely intended for analytical purposes are not within the scope of the journal. Additionally, disciplines such as soil science, polymer science, and metallurgy fall outside the purview of Separation and Purification Technology. Join us in advancing the field of separation and purification methods for sustainable solutions in chemical and environmental engineering.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


