Decorative separator with spongy morphology polyaniline enables dendrite free zinc ion hybrid supercapacitor
IF 5.5
3区 材料科学
Q1 ELECTROCHEMISTRY
引用次数: 0
Abstract
Aqueous zinc-ion hybrid supercapacitors (ZHSCs) have arose as a favourable energy storage device because of their low cost, excellent safety profile, and eco-friendly nature. Unfortunately, the device shorts out due to the glass fiber's strong affinity for zinc ions, which causes zinc dendrites to develop towards the separator. An innovative approach of controlling zinc deposition behaviors through the use of pyrolysis polyaniline functionalized glass fiber (GF-PANI) separator is presented here. Zinc ion flow and deposition behavior are both controlled by the porous PANI structure, which serve to confine the ions. Abundant nitrogen-containing functional groups have the ability to absorb molecules of water. This greatly enhances the zinc anode's reversibility and safety features. Consequently, GF-PANI separators based Zn/Zn symmetric cells have a higher cycle life, lasting up to 800 h at 2 mA cm−2 and 1 mA h cm−2 (120 h). Furthermore, a Zn/JAC cell with GF-PANI separator demonstrates a good specific capacity even after 40,000 cycles at 10 A g−1 in comparison to Zn/GF/JAC cells. This study investigated the design of synergistic, economical, and efficient separators for ZHSCs.

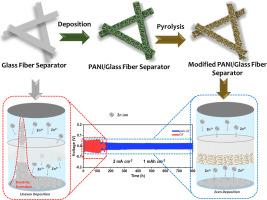
装饰性隔板与海绵状形态聚苯胺使枝晶无锌离子杂化超级电容器
含水锌离子混合超级电容器(ZHSCs)由于其低成本、优异的安全性和生态友好性而成为一种良好的储能装置。不幸的是,由于玻璃纤维对锌离子的亲和力很强,导致锌枝晶向分离器方向发展,导致该装置短路。本文提出了一种利用热解聚苯胺功能化玻璃纤维(GF-PANI)分离器控制锌沉积行为的创新方法。锌离子的流动和沉积行为都受到聚苯胺多孔结构的控制,聚苯胺多孔结构对锌离子起到了限制作用。丰富的含氮官能团具有吸收水分子的能力。这大大提高了锌阳极的可逆性和安全性。因此,基于GF- pani分离器的Zn/Zn对称电池具有更高的循环寿命,在2 mA cm - 2和1 mA h cm - 2 (120 h)下持续长达800小时。此外,与Zn/GF/JAC电池相比,带有GF- pani分离器的Zn/JAC电池即使在10 a g - 1下循环40000次后也表现出良好的比容量。本研究探讨了增效、经济、高效的zhsc分离器的设计。
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文
来源期刊

Electrochimica Acta
工程技术-电化学
CiteScore
11.30
自引率
6.10%
发文量
1634
审稿时长
41 days
期刊介绍:
Electrochimica Acta is an international journal. It is intended for the publication of both original work and reviews in the field of electrochemistry. Electrochemistry should be interpreted to mean any of the research fields covered by the Divisions of the International Society of Electrochemistry listed below, as well as emerging scientific domains covered by ISE New Topics Committee.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


