The auditory midbrain mediates tactile vibration sensing
IF 45.5
1区 生物学
Q1 BIOCHEMISTRY & MOLECULAR BIOLOGY
引用次数: 0
Abstract
Vibrations are ubiquitous in nature, shaping behavior across the animal kingdom. For mammals, mechanical vibrations acting on the body are detected by mechanoreceptors of the skin and deep tissues and processed by the somatosensory system, while sound waves traveling through air are captured by the cochlea and encoded in the auditory system. Here, we report that mechanical vibrations detected by the body’s Pacinian corpuscle neurons, which are distinguished by their ability to entrain to high-frequency (40–1,000 Hz) environmental vibrations, are prominently encoded by neurons in the lateral cortex of the inferior colliculus (LCIC) of the midbrain. Remarkably, most LCIC neurons receive convergent Pacinian and auditory input and respond more strongly to coincident tactile-auditory stimulation than to either modality alone. Moreover, the LCIC is required for behavioral responses to high-frequency mechanical vibrations. Thus, environmental vibrations captured by Pacinian corpuscles are encoded in the auditory midbrain to mediate behavior.
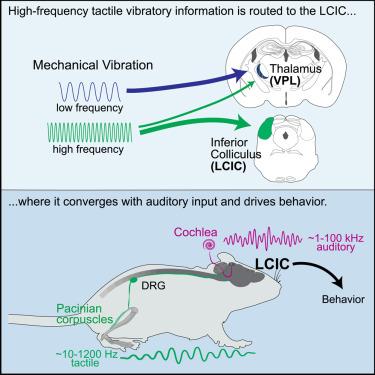
听觉中脑介导触觉振动感知
振动在自然界无处不在,影响着整个动物王国的行为。对于哺乳动物来说,作用于身体的机械振动由皮肤和深层组织的机械感受器检测到,并由体感系统进行处理,而穿过空气的声波则由耳蜗捕捉并编码到听觉系统中。在这里,我们报告了人体帕氏体神经元检测到的机械振动,这些神经元能够与高频率(40-1,000 Hz)的环境振动保持一致,它们主要由中脑下丘外侧皮层(LCIC)的神经元编码。值得注意的是,大多数 LCIC 神经元都能接收帕西尼亚和听觉输入,并且对触觉和听觉同时刺激的反应比单独对其中一种模式刺激的反应更强烈。此外,对高频机械振动的行为反应也需要 LCIC。因此,帕氏体捕捉到的环境振动被编码到听觉中脑,从而介导行为。
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文
来源期刊
Cell
生物-生化与分子生物学
CiteScore
110.00
自引率
0.80%
发文量
396
审稿时长
2 months
期刊介绍:
Cells is an international, peer-reviewed, open access journal that focuses on cell biology, molecular biology, and biophysics. It is affiliated with several societies, including the Spanish Society for Biochemistry and Molecular Biology (SEBBM), Nordic Autophagy Society (NAS), Spanish Society of Hematology and Hemotherapy (SEHH), and Society for Regenerative Medicine (Russian Federation) (RPO).
The journal publishes research findings of significant importance in various areas of experimental biology, such as cell biology, molecular biology, neuroscience, immunology, virology, microbiology, cancer, human genetics, systems biology, signaling, and disease mechanisms and therapeutics. The primary criterion for considering papers is whether the results contribute to significant conceptual advances or raise thought-provoking questions and hypotheses related to interesting and important biological inquiries.
In addition to primary research articles presented in four formats, Cells also features review and opinion articles in its "leading edge" section, discussing recent research advancements and topics of interest to its wide readership.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


