The catalytic role of Cr(VI) in promoting the degradation of organic pollutants via TiO2/SnO2/BiVO4 photoelectrocatalysis: as an electron transfer carrier
IF 7.3
2区 环境科学与生态学
Q1 ENVIRONMENTAL SCIENCES
引用次数: 0
Abstract
Regarding the coexistence of heavy metals and organic pollutants, many studies have achieved synergistic removal of Cr(VI) and organic pollutants under acidic conditions. However, the role of Cr(VI) in the removal of organic pollutants under neutral conditions may have been overlooked. Here, the catalytic effect of Cr(VI) in promoting the photoelectrocatalytic degradation of organic pollutants was investigated using a ternary system, which was a combination of photocatalysis, electrocatalysis and Cr(VI) catalysis. A fabricated TiO2/SnO2/BiVO4 composite was used as an anode for degradation experiments. We found that the degradation efficiency and final removal of rhodamine B (RhB), ofloxacin (OFL), and tetracycline (TC) were improved by increasing the Cr(VI) concentration within the range of 10 to 20 mg L-1. And the addition of Cr(VI) sharply improved the removal of the three organic pollutants under the optimal conditions (from 65% to 96% for RhB, from 34% to 81% for OFL, and from 43% to 100% for TC). According to the results of different processing methods and quenching experiments, we proposed a new catalytic mechanism that, Cr(VI) promotes PEC degradation by acting as an electron transfer carrier between TiO2/SnO2/BiVO4 and O2.
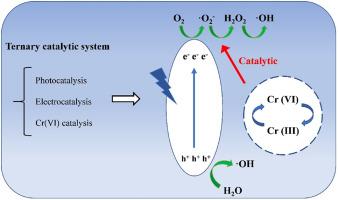
Cr(VI)作为电子转移载体通过TiO2/SnO2/BiVO4光电催化促进有机污染物降解的催化作用
对于重金属与有机污染物共存的问题,许多研究已经在酸性条件下实现了Cr(VI)与有机污染物的协同去除。然而,在中性条件下,Cr(VI)在去除有机污染物中的作用可能被忽视。本文采用光催化、电催化和Cr(VI)催化的三元体系,研究了Cr(VI)促进光电催化降解有机污染物的催化作用。采用制备的TiO2/SnO2/BiVO4复合材料作为阳极进行降解实验。研究发现,在10 ~ 20 mg L-1的Cr(VI)浓度范围内,增加Cr(VI)浓度可提高罗丹明B (RhB)、氧氟沙星(OFL)和四环素(TC)的降解效率和最终去除率。在最佳条件下,Cr(VI)的加入显著提高了三种有机污染物的去除率(RhB从65%提高到96%,OFL从34%提高到81%,TC从43%提高到100%)。根据不同的加工方法和淬火实验结果,我们提出了一种新的催化机制,即Cr(VI)作为TiO2/SnO2/BiVO4和O2之间的电子转移载体促进PEC降解。
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文
来源期刊

Environmental Pollution
环境科学-环境科学
CiteScore
16.00
自引率
6.70%
发文量
2082
审稿时长
2.9 months
期刊介绍:
Environmental Pollution is an international peer-reviewed journal that publishes high-quality research papers and review articles covering all aspects of environmental pollution and its impacts on ecosystems and human health.
Subject areas include, but are not limited to:
• Sources and occurrences of pollutants that are clearly defined and measured in environmental compartments, food and food-related items, and human bodies;
• Interlinks between contaminant exposure and biological, ecological, and human health effects, including those of climate change;
• Contaminants of emerging concerns (including but not limited to antibiotic resistant microorganisms or genes, microplastics/nanoplastics, electronic wastes, light, and noise) and/or their biological, ecological, or human health effects;
• Laboratory and field studies on the remediation/mitigation of environmental pollution via new techniques and with clear links to biological, ecological, or human health effects;
• Modeling of pollution processes, patterns, or trends that is of clear environmental and/or human health interest;
• New techniques that measure and examine environmental occurrences, transport, behavior, and effects of pollutants within the environment or the laboratory, provided that they can be clearly used to address problems within regional or global environmental compartments.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


