Chronic polyaluminum chloride perturbation shaped community assembly patterns: An overlooked mechanism of nitrogen removal inhibition
IF 13.3
1区 工程技术
Q1 ENGINEERING, CHEMICAL
引用次数: 0
Abstract
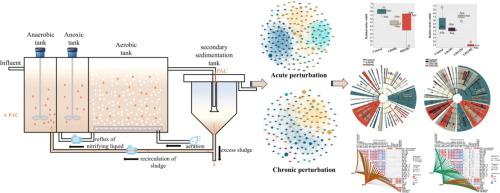
Prior research indicated that aluminum ions (Al3+) could disrupt iron homeostasis in cells in synchronous biological nutrient removal and aluminum-based chemical phosphorus removal (CPR). However, the underlying microbial community assembly mechanisms in response to chronic polyaluminum chloride (PAC) disturbance remained unclear. Herein, post-precipitation experiments were conducted with PAC at varying concentrations (0, 40, 80 mg/L) in continuous flow anaerobic-anoxic–oxic systems over a 240-day period. Phosphorus was mainly removed through chemical processes, with minimal changes in polyphosphate accumulating organisms (PAOs) abundance and metabolic genes for polyphosphate and poly-β-hydroxybutyrate. Chemical phosphorus precipitates accounted for 74.7 ∼ 79.3 % in the activated sludge, with aluminum- and iron-phosphorus compounds (AlPs and FePs) being the primary components. Under low-level PAC, the average total inorganic nitrogen (TIN) removal efficiency shifted from short-term enhancement (≈73 %) to long-term inhibition (<60 %), with the inhibition being more pronounced than the acute disturbance caused by high-level PAC (≈63.5 %). Nitrification was most vulnerable to damage compared to denitrification under PAC stress. Although Al3+ initially replaced cellular iron, the iron-induced autotrophic denitrifying bacteria (Azospira) or iron-reducing bacteria (Trichococcus) became dominant, thereby compensating for damage to the tricarboxylic acid cycle and electron transfer. Upon the accumulation of Al3+ reaching 100 mg/g, the ecosystem achieved a new state of stability. Acute PAC disturbance enhanced community cooperation and facilitated rapid network reconfiguration, whereas chronic disturbance resulted in the loss of species and a reduction in network complexity. PAC altered the dynamics of nitrogen conversion by modifying electron flow, with lower concentrations augmenting denitrification and higher concentrations detrimental to nitrification. The biosystem adapted to low PAC levels by rerouting electrons around complex IV, which enhanced electron flow efficiency; however, this adaptation did not preserve in chronic disturbance. This study elucidates microbial assembly mechanisms under varying PAC disturbances, providing insights into the ecological resilience and functional stability of wastewater treatment systems.
长期多氯化铝扰动塑造了群落组装模式:被忽视的脱氮抑制机制
先前的研究表明,铝离子(Al3+)在同步生物除营养物和铝基化学除磷(CPR)过程中会破坏细胞内铁的稳态。然而,对慢性聚合氯化铝(PAC)干扰的潜在微生物群落组装机制尚不清楚。在此,沉淀后实验采用不同浓度的PAC(0、40、80 mg/L)在厌氧-缺氧-缺氧连续流动系统中进行,持续240天。磷主要通过化学过程去除,聚磷酸盐积累生物(PAOs)丰度和聚磷酸盐和聚β-羟基丁酸盐代谢基因的变化很小。活性污泥中化学磷沉淀占74.7 ~ 79.3 %,其中铝和铁磷化合物(AlPs和FePs)是主要成分。低浓度PAC对总无机氮(TIN)的平均去除效率由短期增强(≈73 %)向长期抑制(<60 %)转变,且抑制作用比高浓度PAC引起的急性干扰(≈63.5 %)更为明显。在PAC胁迫下,硝化作用比反硝化作用更容易受到破坏。虽然Al3+最初取代了细胞铁,但铁诱导的自养反硝化细菌(Azospira)或铁还原细菌(Trichococcus)成为主导,从而补偿了对三羧酸循环和电子转移的损害。当Al3+积累量达到100 mg/g时,生态系统达到了新的稳定状态。急性PAC干扰增强了群落合作,促进了网络的快速重构,而慢性干扰导致物种的损失和网络复杂性的降低。PAC通过改变电子流改变氮转化动力学,低浓度增强反硝化作用,高浓度不利于硝化作用。生物系统通过改变电子在络合物IV周围的路径来适应低PAC水平,从而提高了电子流效率;然而,这种适应并没有在慢性干扰中保持。本研究阐明了不同PAC干扰下的微生物组装机制,为污水处理系统的生态弹性和功能稳定性提供了见解。
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文
来源期刊

Chemical Engineering Journal
工程技术-工程:化工
CiteScore
21.70
自引率
9.30%
发文量
6781
审稿时长
2.4 months
期刊介绍:
The Chemical Engineering Journal is an international research journal that invites contributions of original and novel fundamental research. It aims to provide an international platform for presenting original fundamental research, interpretative reviews, and discussions on new developments in chemical engineering. The journal welcomes papers that describe novel theory and its practical application, as well as those that demonstrate the transfer of techniques from other disciplines. It also welcomes reports on carefully conducted experimental work that is soundly interpreted. The main focus of the journal is on original and rigorous research results that have broad significance. The Catalysis section within the Chemical Engineering Journal focuses specifically on Experimental and Theoretical studies in the fields of heterogeneous catalysis, molecular catalysis, and biocatalysis. These studies have industrial impact on various sectors such as chemicals, energy, materials, foods, healthcare, and environmental protection.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


