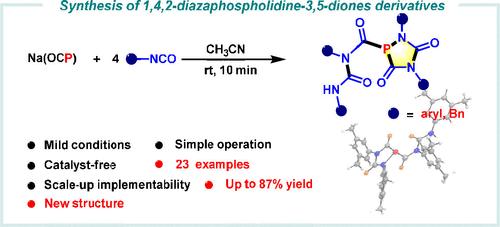
Synthesis of 1,4,2-Diazaphospholidine-3,5-diones Using Na(OCP) as the “P” Source
IF 3.3
2区 化学
Q1 CHEMISTRY, ORGANIC
引用次数: 0
Abstract
A refined synthesis of 1,4,2-diazaphospholidine-3,5-dione derivatives was achieved through a cyclization reaction involving Na(OCP) and isocyanates. Na(OCP) was demonstrated to be a relatively stable and safe source of phosphorus, enabling the production of diverse 1,4,2-diazaphospholidine-3,5-dione derivatives with high yields. The reaction proceeds efficiently under catalyst-free and mild conditions. Both experimental findings and density functional theory calculations have elucidated that the process involves a crucial step of carbon monoxide elimination, which provides deeper insight into the reaction mechanism.
求助全文
约1分钟内获得全文
求助全文
来源期刊

Journal of Organic Chemistry
化学-有机化学
CiteScore
6.20
自引率
11.10%
发文量
1467
审稿时长
2 months
期刊介绍:
Journal of Organic Chemistry welcomes original contributions of fundamental research in all branches of the theory and practice of organic chemistry. In selecting manuscripts for publication, the editors place emphasis on the quality and novelty of the work, as well as the breadth of interest to the organic chemistry community.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


