Insights into the Roles of Different Iron Species on Zeolites for N2O Selective Catalytic Reduction by CO
IF 10.8
1区 环境科学与生态学
Q1 ENGINEERING, ENVIRONMENTAL
引用次数: 0
Abstract
Iron zeolites are promising candidates for mitigating nitrous oxide (N2O), a potent greenhouse gas and contributor to stratospheric ozone destruction. However, the atomic-level mechanisms by which different iron species, including isolated sites, clusters, and particles, participate in N2O decomposition in the presence of CO still remain poorly understood, which hinders the application of the reaction in practical technology. Herein, through experiments and density functional theory (DFT) calculations, we identified that isolated iron sites were active for N2O activation to generate adsorbed O* species, which readily reacted with CO following the Eley–Rideal (E-R) mechanism. In contrast, Fe2O3 particles exhibited a different reaction pathway, directly reacting with CO to generate oxygen vacancies (Ov), which could efficiently dissociate N2O following the Mars-van Krevelen (MvK) mechanism. Moreover, the transformation of iron oxide clusters into undercoordinated FeOx species by CO was also revealed through various techniques, such as CO-temperature-programmed reduction (TPR), and ab initio molecular dynamics (AIMD) simulations. Our study provides deeper insights into the roles of different iron species in N2O-SCR by CO, and is anticipated to facilitate the understanding of multicomponent catalysis and the design of efficient iron-containing catalysts for practical applications.
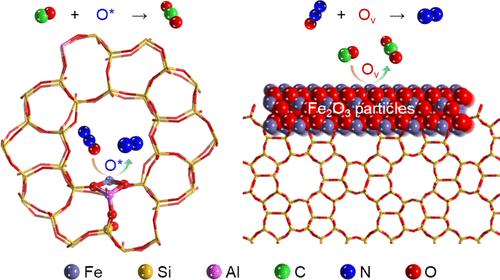
不同铁种在沸石中CO选择性催化还原N2O作用的研究
铁沸石是缓解一氧化二氮(N2O)的有希望的候选者,N2O是一种强有力的温室气体,对平流层臭氧的破坏有贡献。然而,在CO存在的情况下,不同铁种类(包括孤立的位点、团簇和颗粒)参与N2O分解的原子水平机制仍然知之甚少,这阻碍了该反应在实际技术中的应用。本文中,通过实验和密度泛函理论(DFT)计算,我们发现孤立的铁位点对N2O活化具有活性,从而产生吸附的O*物质,这些O*物质很容易按照eely - rideal (E-R)机制与CO发生反应。相比之下,Fe2O3颗粒表现出不同的反应途径,直接与CO反应生成氧空位(Ov),可以根据Mars-van Krevelen (MvK)机制有效解离N2O。此外,通过CO-温度程序还原(TPR)和从头算分子动力学(AIMD)模拟等各种技术,也揭示了CO将氧化铁团簇转化为欠配位FeOx物质的过程。本研究对不同铁元素在CO催化N2O-SCR中的作用提供了更深入的认识,并有望促进对多组分催化的理解和实际应用中高效含铁催化剂的设计。
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文
来源期刊

环境科学与技术
环境科学-工程:环境
CiteScore
17.50
自引率
9.60%
发文量
12359
审稿时长
2.8 months
期刊介绍:
Environmental Science & Technology (ES&T) is a co-sponsored academic and technical magazine by the Hubei Provincial Environmental Protection Bureau and the Hubei Provincial Academy of Environmental Sciences.
Environmental Science & Technology (ES&T) holds the status of Chinese core journals, scientific papers source journals of China, Chinese Science Citation Database source journals, and Chinese Academic Journal Comprehensive Evaluation Database source journals. This publication focuses on the academic field of environmental protection, featuring articles related to environmental protection and technical advancements.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


