Ruthenium-complex-catalysed de-ammonification polycondensation of aromatic diamines†
IF 4.1
2区 化学
Q2 POLYMER SCIENCE
引用次数: 0
Abstract
This study explores a pioneering catalytic reaction to obtain functional polymers and valuable byproducts. Using RuCl2(PPh3)3 as a catalyst activates the C–N and N–H bonds in aromatic diamines, when combined with 1,4-butynediol. This activation initiates de-ammonification polycondensation, resulting in aromatic polyamines with a pyrrolyl end group and ammonia as a byproduct. The ammonia generated during the polycondensation process can be captured in cold water. The generation of ammonia during polycondensation was confirmed by UV-vis spectroscopy using the Nessler's reagent method. Subsequently, the aromatic polyamines were further functionalised via polymer reactions with 1,4-butanesultone and butyl isocyanate in the NH group. This yielded products with pendant sodium N-butylsulfonate and N-butylamide groups, respectively. The former exhibited a single-ion conductivity. Potential reaction mechanisms involving Ru-catalysed N–H and C–N bond activation in AD, along with the formation of terminal pyrrolyl groups, were investigated using density functional theorycalculations and 2H NMR spectroscopy.
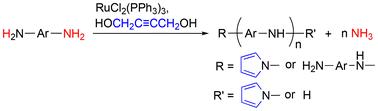

钌络合物催化芳香二胺脱氨缩聚反应
本研究探索了一种开创性的催化反应,以获得功能性聚合物和有价值的副产物。以RuCl2(PPh3)3为催化剂,与1,4-丁炔二醇结合,激活芳香二胺中的C-N和N-H键。这种活化引发脱氨化缩聚,产生具有吡咯基的芳香多胺和氨作为副产物。缩聚过程中产生的氨可以在冷水中捕获。用奈斯勒试剂法用紫外-可见光谱法证实了缩聚过程中氨的生成。随后,芳香族多胺通过与NH基团的1,4-丁烷磺酸和异氰酸丁酯的聚合物反应进一步功能化。该方法得到的产物分别含有n -丁基磺酸钠和n -丁酰胺基团。前者表现出单离子电导率。利用密度泛函理论计算和2H核磁共振波谱研究了AD中钌催化的N-H和C-N键活化以及末端吡啶基形成的潜在反应机制。
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文
来源期刊

Polymer Chemistry
POLYMER SCIENCE-
CiteScore
8.60
自引率
8.70%
发文量
535
审稿时长
1.7 months
期刊介绍:
Polymer Chemistry welcomes submissions in all areas of polymer science that have a strong focus on macromolecular chemistry. Manuscripts may cover a broad range of fields, yet no direct application focus is required.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


