Probing the Biomolecular Interactions of DNA/HSA with the New Sn(IV) Complex and Computational Perspectives: Design, Synthesis, Characterization, Anticancer Activity, and Molecular Modeling Approach
IF 6.8
1区 医学
Q1 CHEMISTRY, MEDICINAL
引用次数: 0
Abstract
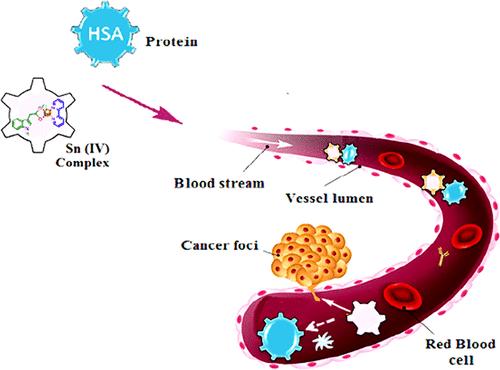
The ligands 2,2′-bipyridyl and indole-3-carboxylic acid were used to create a Sn(IV) complex, which was then synthesized and carefully characterized using elemental analysis and spectroscopic techniques (UV–vis, IR, 1H, 13C, and 119Sn NMR, and ESI-MS) and RXPD. Utilizing biophysical techniques such as UV–vis, fluorescence titrations, circular dichroism, FTIR (for HSA), and cleavage activity (for DNA), in vitro binding studies of Sn(IV) complex and DNA/HSA were satisfied with the strong electrostatic binding interaction of the Sn(IV) complex via the phosphate backbone of the DNA helix as well as in the subdomain IIA of HSA. The observed trend in the binding interactions and computational studies of the Sn(IV) complex was attributed to the nature of the ligands bound to the Sn(IV) center that influences their in vitro activities. The Sn(IV) complex showed sufficient effectiveness to be considered a viable candidate for the creation of anticancer medications.
探索DNA/HSA与新Sn(IV)配合物的生物分子相互作用及其计算视角:设计、合成、表征、抗癌活性和分子建模方法
利用配体2,2′-联吡啶和吲哚-3-羧酸合成了Sn(IV)配合物,并利用元素分析和光谱技术(UV-vis、IR、1H、13C和119Sn NMR以及ESI-MS)和RXPD对其进行了仔细的表征。利用生物物理技术,如紫外-可见、荧光滴定、圆二色性、FTIR(用于HSA)和切割活性(用于DNA),对Sn(IV)配合物和DNA/HSA的体外结合研究表明,Sn(IV)配合物通过DNA螺旋的磷酸主链以及HSA的子结构域IIA进行了强静电结合。Sn(IV)配合物的结合相互作用和计算研究中观察到的趋势归因于与Sn(IV)中心结合的配体的性质,这影响了它们的体外活性。Sn(IV)复合物显示出足够的有效性,被认为是创造抗癌药物的可行候选者。
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文
来源期刊

Journal of Medicinal Chemistry
医学-医药化学
CiteScore
4.00
自引率
11.00%
发文量
804
审稿时长
1.9 months
期刊介绍:
The Journal of Medicinal Chemistry is a prestigious biweekly peer-reviewed publication that focuses on the multifaceted field of medicinal chemistry. Since its inception in 1959 as the Journal of Medicinal and Pharmaceutical Chemistry, it has evolved to become a cornerstone in the dissemination of research findings related to the design, synthesis, and development of therapeutic agents.
The Journal of Medicinal Chemistry is recognized for its significant impact in the scientific community, as evidenced by its 2022 impact factor of 7.3. This metric reflects the journal's influence and the importance of its content in shaping the future of drug discovery and development. The journal serves as a vital resource for chemists, pharmacologists, and other researchers interested in the molecular mechanisms of drug action and the optimization of therapeutic compounds.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


