Regulation of human interferon signaling by transposon exonization
IF 45.5
1区 生物学
Q1 BIOCHEMISTRY & MOLECULAR BIOLOGY
引用次数: 0
Abstract
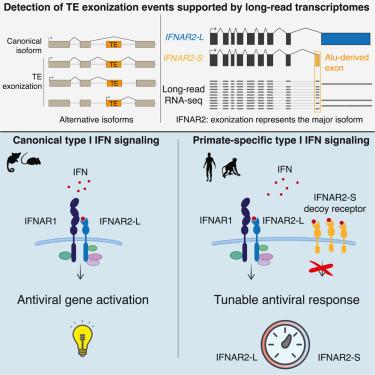
Innate immune signaling is essential for clearing pathogens and damaged cells and must be tightly regulated to avoid excessive inflammation or autoimmunity. Here, we found that the alternative splicing of exons derived from transposable elements is a key mechanism controlling immune signaling in human cells. By analyzing long-read transcriptome datasets, we identified numerous transposon exonization events predicted to generate functional protein variants of immune genes, including the type I interferon receptor IFNAR2. We demonstrated that the transposon-derived isoform of IFNAR2 is more highly expressed than the canonical isoform in almost all tissues and functions as a decoy receptor that potently inhibits interferon signaling, including in cells infected with severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2). Our findings uncover a primate-specific axis controlling interferon signaling and show how a transposon exonization event can be co-opted for immune regulation.
转座子外显子对人干扰素信号的调控
先天免疫信号对于清除病原体和受损细胞至关重要,必须严格调节以避免过度炎症或自身免疫。在这里,我们发现来自转座因子的外显子的选择性剪接是控制人类细胞免疫信号的关键机制。通过分析长读转录组数据集,我们发现了许多转座子外显子事件,预测会产生免疫基因的功能蛋白变异,包括I型干扰素受体IFNAR2。我们证明,IFNAR2的转座子衍生异构体在几乎所有组织中比典型异构体表达更高,并作为诱骗受体有效抑制干扰素信号传导,包括在感染了严重急性呼吸综合征冠状病毒2 (SARS-CoV-2)的细胞中。我们的发现揭示了灵长类特异性轴控制干扰素信号,并展示了转座子外显子事件如何被免疫调节所利用。
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文
来源期刊
Cell
生物-生化与分子生物学
CiteScore
110.00
自引率
0.80%
发文量
396
审稿时长
2 months
期刊介绍:
Cells is an international, peer-reviewed, open access journal that focuses on cell biology, molecular biology, and biophysics. It is affiliated with several societies, including the Spanish Society for Biochemistry and Molecular Biology (SEBBM), Nordic Autophagy Society (NAS), Spanish Society of Hematology and Hemotherapy (SEHH), and Society for Regenerative Medicine (Russian Federation) (RPO).
The journal publishes research findings of significant importance in various areas of experimental biology, such as cell biology, molecular biology, neuroscience, immunology, virology, microbiology, cancer, human genetics, systems biology, signaling, and disease mechanisms and therapeutics. The primary criterion for considering papers is whether the results contribute to significant conceptual advances or raise thought-provoking questions and hypotheses related to interesting and important biological inquiries.
In addition to primary research articles presented in four formats, Cells also features review and opinion articles in its "leading edge" section, discussing recent research advancements and topics of interest to its wide readership.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


