Nickel-Based Catalysts for Dry Reforming of Methane in the Absence/Presence of H2S: Effect of SiO2 and Cu
IF 3.8
3区 工程技术
Q2 ENGINEERING, CHEMICAL
引用次数: 0
Abstract
Dry reforming of methane (DRM) can utilize methane and carbon dioxide to produce synthesis gas. The nickel-based catalysts used in the DRM are easily poisoned by H2S in the feed gas. This study evaluated the effect of SiO2 and Cu on the Ni/Al2O3 catalyst during DRM in the absence/presence of H2S. XRD, N2 adsorption/desorption, TEM, XPS, H2-TPR, CO2-TPD, and TGA were used to characterize the catalysts. The results show that in the absence of H2S, adding SiO2 can effectively reduce the amount of coke on the catalyst surface, whereas adding Cu can effectively change the nature of coke to more reactive filament carbon. The presence of H2S facilitates coke deposition on the catalyst surface. The synergistic effect of strengthening the metal–support interaction by SiO2 and absorbing H2S by Cu results in simultaneous coke and sulfur resistance of the NiAl1Si3Cu catalyst.
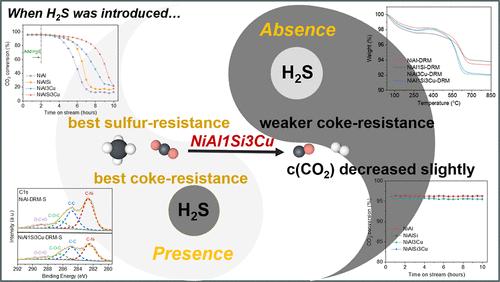
无/有H2S条件下甲烷干重整镍基催化剂:SiO2和Cu的影响
甲烷干式重整(DRM)是利用甲烷和二氧化碳生产合成气。在DRM中使用的镍基催化剂容易被原料气中的H2S毒害。本研究评估了在H2S存在或不存在的情况下,SiO2和Cu对Ni/Al2O3催化剂DRM的影响。采用XRD、N2吸附/解吸、TEM、XPS、H2-TPR、CO2-TPD和TGA对催化剂进行了表征。结果表明,在没有H2S的情况下,添加SiO2可以有效地减少催化剂表面的焦炭数量,而添加Cu可以有效地改变焦炭的性质,使其变成活性更强的长丝碳。H2S的存在有利于焦炭在催化剂表面的沉积。SiO2增强金属-载体相互作用和Cu吸附H2S的协同效应导致NiAl1Si3Cu催化剂同时具有抗焦性和抗硫性。
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文
来源期刊

Industrial & Engineering Chemistry Research
工程技术-工程:化工
CiteScore
7.40
自引率
7.10%
发文量
1467
审稿时长
2.8 months
期刊介绍:
ndustrial & Engineering Chemistry, with variations in title and format, has been published since 1909 by the American Chemical Society. Industrial & Engineering Chemistry Research is a weekly publication that reports industrial and academic research in the broad fields of applied chemistry and chemical engineering with special focus on fundamentals, processes, and products.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


