Structure-Based Discovery of a Series of Covalent, Orally Bioavailable, and Selective BFL1 Inhibitors
IF 6.8
1区 医学
Q1 CHEMISTRY, MEDICINAL
引用次数: 0
Abstract
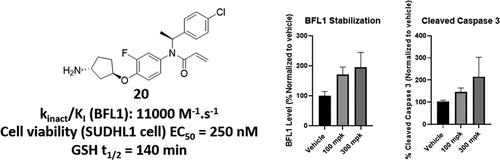
BFL1, a member of the antiapoptotic BCL2 family, has been relatively understudied compared to its counterparts despite evidence of its overexpression in various hematological malignancies. Across two articles, we describe the development of BFL1 in vivo tools. The first article describes the hit identification from a covalent fragment library and the subsequent evolution from the hit to compound 6.22 This work reports the structure-based optimization of compound 6 into a series of BFL1 inhibitors selective over the other BCL2 family members, with low nanomolar cellular activity when combined with AZD5991, exemplified by compound 20. Compound 20 demonstrated a cell death phenotype in SUDHL1 and OCILY10 cell lines and in the in vivo study, BFL1 stabilization and cleaved caspase 3 activation were observed in a dose-dependent manner. In addition, the enzymatic turnover studies with the BFL1 protein showed that compound 20 stabilized the protein, extending the half-life to 10.8 h.
求助全文
约1分钟内获得全文
求助全文
来源期刊

Journal of Medicinal Chemistry
医学-医药化学
CiteScore
4.00
自引率
11.00%
发文量
804
审稿时长
1.9 months
期刊介绍:
The Journal of Medicinal Chemistry is a prestigious biweekly peer-reviewed publication that focuses on the multifaceted field of medicinal chemistry. Since its inception in 1959 as the Journal of Medicinal and Pharmaceutical Chemistry, it has evolved to become a cornerstone in the dissemination of research findings related to the design, synthesis, and development of therapeutic agents.
The Journal of Medicinal Chemistry is recognized for its significant impact in the scientific community, as evidenced by its 2022 impact factor of 7.3. This metric reflects the journal's influence and the importance of its content in shaping the future of drug discovery and development. The journal serves as a vital resource for chemists, pharmacologists, and other researchers interested in the molecular mechanisms of drug action and the optimization of therapeutic compounds.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


