An Improved Commercial Process for the Preparation of Lifitegrast
IF 3.5
3区 化学
Q2 CHEMISTRY, APPLIED
引用次数: 0
Abstract
A straightforward, efficient, and scalable commercial manufacturing process was developed for the ophthalmic anti-inflammatory drug lifitegrast via a novel ester intermediate from commercially available starting materials. Lifitegrast (Xiidra) was approved by the FDA on July 11, 2016, for the treatment of signs and symptoms of dry eye, a syndrome called keratoconjunctivitis sicca. The breakthrough step of this new process is the discovery of an N-Boc deprotection reaction that simultaneously transesterifies an intermediate to a new ester by using oxalyl chloride, which has favorable isolation properties. As a result of transesterification, the hydrolysis of the new ester intermediate occurs under milder conditions, which improves the quality of the product by reducing racemization. Lifitegrast prepared from this new process complied with the quality guidelines, as per the International Council for Harmonization (ICH). By using this new process, lifitegrast was produced on a 2 kg scale with an overall yield of 66%.
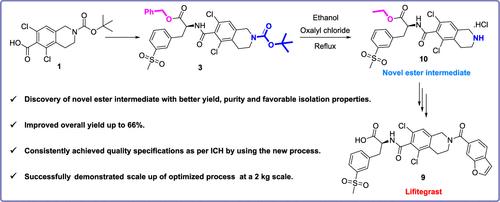
一种改进的lifitgrast制备工艺
通过一种新型酯中间体,从市售原料中提取眼部消炎药lifitegrast,开发了一种简单、高效、可扩展的商业生产工艺。Lifitegrast (Xiidra)于2016年7月11日获得FDA批准,用于治疗干眼症的体征和症状,这是一种称为干燥性角膜结膜炎的综合征。该新工艺的突破性进展是发现了一种N-Boc脱保护反应,该反应利用草酰氯将中间体同时酯交换成新的酯,具有良好的分离性能。由于酯交换反应,新酯中间体的水解在较温和的条件下发生,从而通过减少外消旋作用提高了产品的质量。按照国际协调理事会(ICH)的要求,新工艺制备的lifitgrast符合质量准则。采用该新工艺,可生产2公斤的lifitgrast,总收率为66%。
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文
来源期刊
CiteScore
6.90
自引率
14.70%
发文量
251
审稿时长
2 months
期刊介绍:
The journal Organic Process Research & Development serves as a communication tool between industrial chemists and chemists working in universities and research institutes. As such, it reports original work from the broad field of industrial process chemistry but also presents academic results that are relevant, or potentially relevant, to industrial applications. Process chemistry is the science that enables the safe, environmentally benign and ultimately economical manufacturing of organic compounds that are required in larger amounts to help address the needs of society. Consequently, the Journal encompasses every aspect of organic chemistry, including all aspects of catalysis, synthetic methodology development and synthetic strategy exploration, but also includes aspects from analytical and solid-state chemistry and chemical engineering, such as work-up tools,process safety, or flow-chemistry. The goal of development and optimization of chemical reactions and processes is their transfer to a larger scale; original work describing such studies and the actual implementation on scale is highly relevant to the journal. However, studies on new developments from either industry, research institutes or academia that have not yet been demonstrated on scale, but where an industrial utility can be expected and where the study has addressed important prerequisites for a scale-up and has given confidence into the reliability and practicality of the chemistry, also serve the mission of OPR&D as a communication tool between the different contributors to the field.

 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


