Ageing limits stemness and tumorigenesis by reprogramming iron homeostasis
IF 50.5
1区 综合性期刊
Q1 MULTIDISCIPLINARY SCIENCES
引用次数: 0
Abstract
Ageing is associated with a decline in the number and fitness of adult stem cells1,2. Ageing-associated loss of stemness is posited to suppress tumorigenesis3,4, but this hypothesis has not been tested in vivo. Here we use physiologically aged autochthonous genetically engineered5,6 mouse models and primary cells5,6 to demonstrate that ageing suppresses lung cancer initiation and progression by degrading the stemness of the alveolar cell of origin. This phenotype is underpinned by the ageing-associated induction of the transcription factor NUPR1 and its downstream target lipocalin-2 in the cell of origin in mice and humans, which leads to functional iron insufficiency in the aged cells. Genetic inactivation of the NUPR1–lipocalin-2 axis or iron supplementation rescues stemness and promotes the tumorigenic potential of aged alveolar cells. Conversely, targeting the NUPR1–lipocalin-2 axis is detrimental to young alveolar cells through ferroptosis induction. Ageing-associated DNA hypomethylation at specific enhancer sites is associated with increased NUPR1 expression, which is recapitulated in young alveolar cells through DNA methylation inhibition. We uncover that ageing drives functional iron insufficiency that leads to loss of stemness and tumorigenesis but promotes resistance to ferroptosis. These findings have implications for the therapeutic modulation of cellular iron homeostasis in regenerative medicine and in cancer prevention. Furthermore, our findings are consistent with a model whereby most human cancers initiate at a young age, thereby highlighting the importance of directing cancer prevention efforts towards young individuals. Studies using mouse models of lung adenocarcinoma identify an association between age, iron homeostasis and tumour initiation potential that involves NUPR1 and lipocalin-2.
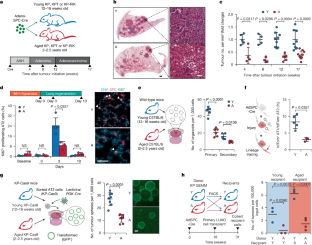

衰老通过重编程铁稳态来限制干细胞和肿瘤的发生
衰老与成体干细胞数量和适应性的下降有关1,2。衰老相关的干性丧失被认为可以抑制肿瘤发生3,4,但这一假设尚未在体内得到验证。在这里,我们使用生理衰老的原生基因工程小鼠模型和原代细胞5,6来证明衰老通过降低肺泡细胞起源的干性来抑制肺癌的发生和进展。这种表型是由小鼠和人类细胞中与衰老相关的转录因子NUPR1及其下游靶点脂钙素-2的诱导所支持的,这导致衰老细胞中的功能性铁不足。基因失活NUPR1-lipocalin-2轴或补铁可挽救干细胞并促进衰老肺泡细胞的致瘤潜能。相反,靶向NUPR1-lipocalin-2轴通过诱导铁下垂对年轻肺泡细胞有害。特定增强子位点的衰老相关DNA低甲基化与NUPR1表达增加相关,这在年轻的肺泡细胞中通过DNA甲基化抑制得以重现。我们发现,衰老驱动功能性铁不足,导致干性丧失和肿瘤发生,但促进对铁下垂的抵抗。这些发现对再生医学和癌症预防中细胞铁稳态的治疗性调节具有启示意义。此外,我们的发现与大多数人类癌症在年轻时开始的模型一致,从而强调了将癌症预防工作指导给年轻人的重要性。
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文
来源期刊

Nature
综合性期刊-综合性期刊
CiteScore
90.00
自引率
1.20%
发文量
3652
审稿时长
3 months
期刊介绍:
Nature is a prestigious international journal that publishes peer-reviewed research in various scientific and technological fields. The selection of articles is based on criteria such as originality, importance, interdisciplinary relevance, timeliness, accessibility, elegance, and surprising conclusions. In addition to showcasing significant scientific advances, Nature delivers rapid, authoritative, insightful news, and interpretation of current and upcoming trends impacting science, scientists, and the broader public. The journal serves a dual purpose: firstly, to promptly share noteworthy scientific advances and foster discussions among scientists, and secondly, to ensure the swift dissemination of scientific results globally, emphasizing their significance for knowledge, culture, and daily life.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


