In vivo affinity maturation of the CD4 domains of an HIV-1-entry inhibitor
IF 26.8
1区 医学
Q1 ENGINEERING, BIOMEDICAL
引用次数: 0
Abstract
Human proteins repurposed as biologics for clinical use have been engineered through in vitro techniques that improve the affinity of the biologics for their ligands. However, the techniques do not select against properties, such as protease sensitivity or self-reactivity, that impair the biologics’ clinical efficacy. Here we show that the B-cell receptors of primary murine B cells can be engineered to affinity mature in vivo the human CD4 domains of the HIV-1-entry inhibitor CD4 immunoadhesin (CD4-Ig). Specifically, we introduced genes encoding the CD4 domains 1 and 2 (D1D2) of a half-life-enhanced form of CD4-Ig (CD4-Ig-v0) into the heavy-chain loci of murine B cells and adoptively transferred these cells into wild-type mice. After immunization, the B cells proliferated, class switched, affinity matured and produced D1D2-presenting antibodies. Somatic hypermutations in the D1D2-encoding region of the engrafted cells improved the binding affinity of CD4-Ig-v0 for the HIV-1 envelope glycoprotein and the inhibitor’s ability to neutralize a panel of HIV-1 isolates without impairing its pharmacokinetic properties. In vivo affinity maturation of non-antibody protein biologics may guide the development of more effective therapeutics. The B-cell receptor of primary mouse B cells can be engineered to affinity mature an HIV-1-entry inhibitor in vivo.
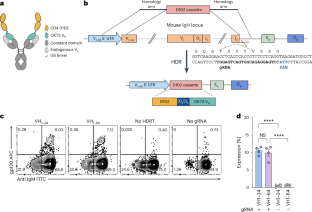

hiv -1进入抑制剂CD4结构域的体内亲和成熟
人类蛋白质作为临床使用的生物制剂已经通过体外技术进行了改造,提高了生物制剂与其配体的亲和力。然而,这些技术并没有选择损害生物制剂临床疗效的特性,如蛋白酶敏感性或自反应性。本研究表明,原代小鼠B细胞的B细胞受体可以被改造成在体内与hiv -1进入抑制剂CD4免疫粘附素(CD4- ig)的人CD4区域亲和成熟。具体来说,我们将编码半衰期增强形式CD4- ig (CD4- ig -v0)的CD4结构域1和2 (D1D2)的基因引入小鼠B细胞的重链位点,并过继地将这些细胞转移到野生型小鼠中。免疫后,B细胞增殖、切换类、亲和成熟并产生d1d2呈递抗体。植入细胞的d1d2编码区的体细胞超突变改善了CD4-Ig-v0对HIV-1包膜糖蛋白的结合亲和力,以及抑制剂中和一组HIV-1分离株而不损害其药代动力学特性的能力。非抗体蛋白生物制剂的体内亲和成熟可以指导更有效治疗方法的开发。
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文
来源期刊

Nature Biomedical Engineering
Medicine-Medicine (miscellaneous)
CiteScore
45.30
自引率
1.10%
发文量
138
期刊介绍:
Nature Biomedical Engineering is an online-only monthly journal that was launched in January 2017. It aims to publish original research, reviews, and commentary focusing on applied biomedicine and health technology. The journal targets a diverse audience, including life scientists who are involved in developing experimental or computational systems and methods to enhance our understanding of human physiology. It also covers biomedical researchers and engineers who are engaged in designing or optimizing therapies, assays, devices, or procedures for diagnosing or treating diseases. Additionally, clinicians, who make use of research outputs to evaluate patient health or administer therapy in various clinical settings and healthcare contexts, are also part of the target audience.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


