Opportunities and challenges of single-cell and spatially resolved genomics methods for neuroscience discovery
IF 21.2
1区 医学
Q1 NEUROSCIENCES
引用次数: 0
Abstract
Over the past decade, single-cell genomics technologies have allowed scalable profiling of cell-type-specific features, which has substantially increased our ability to study cellular diversity and transcriptional programs in heterogeneous tissues. Yet our understanding of mechanisms of gene regulation or the rules that govern interactions between cell types is still limited. The advent of new computational pipelines and technologies, such as single-cell epigenomics and spatially resolved transcriptomics, has created opportunities to explore two new axes of biological variation: cell-intrinsic regulation of cell states and expression programs and interactions between cells. Here, we summarize the most promising and robust technologies in these areas, discuss their strengths and limitations and discuss key computational approaches for analysis of these complex datasets. We highlight how data sharing and integration, documentation, visualization and benchmarking of results contribute to transparency, reproducibility, collaboration and democratization in neuroscience, and discuss needs and opportunities for future technology development and analysis. This review provides an overview of analysis and experimental design of single-cell omics in the brain, emphasizing epigenomics and spatial omics. The authors discuss how the computational and experimental designs are interlinked, with both being guided by the biological questions.
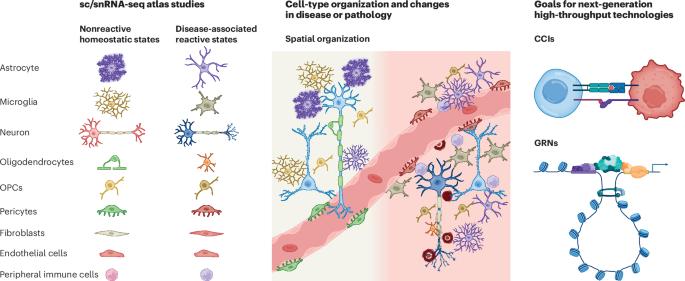
单细胞和空间分辨基因组学方法在神经科学发现中的机遇和挑战
在过去的十年中,单细胞基因组学技术已经允许对细胞类型特异性特征进行可扩展的分析,这大大提高了我们研究异质组织中细胞多样性和转录程序的能力。然而,我们对基因调控机制或控制细胞类型之间相互作用的规则的理解仍然有限。新的计算管道和技术的出现,如单细胞表观基因组学和空间解析转录组学,为探索生物变异的两个新轴创造了机会:细胞状态和表达程序的细胞内在调节以及细胞之间的相互作用。在这里,我们总结了这些领域中最有前途和最强大的技术,讨论了它们的优势和局限性,并讨论了分析这些复杂数据集的关键计算方法。我们强调数据共享和集成、文档化、可视化和结果基准化如何有助于神经科学的透明度、可重复性、协作性和民主化,并讨论未来技术发展和分析的需求和机会。本文综述了脑单细胞组学的分析和实验设计,重点介绍了表观基因组学和空间组学。作者讨论了计算设计和实验设计是如何相互联系的,两者都受到生物学问题的指导。
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文
来源期刊

Nature neuroscience
医学-神经科学
CiteScore
38.60
自引率
1.20%
发文量
212
审稿时长
1 months
期刊介绍:
Nature Neuroscience, a multidisciplinary journal, publishes papers of the utmost quality and significance across all realms of neuroscience. The editors welcome contributions spanning molecular, cellular, systems, and cognitive neuroscience, along with psychophysics, computational modeling, and nervous system disorders. While no area is off-limits, studies offering fundamental insights into nervous system function receive priority.
The journal offers high visibility to both readers and authors, fostering interdisciplinary communication and accessibility to a broad audience. It maintains high standards of copy editing and production, rigorous peer review, rapid publication, and operates independently from academic societies and other vested interests.
In addition to primary research, Nature Neuroscience features news and views, reviews, editorials, commentaries, perspectives, book reviews, and correspondence, aiming to serve as the voice of the global neuroscience community.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


