4-Substituted-2-Thiazole Amides as Viral Replication Inhibitors of Alphaviruses
IF 6.8
1区 医学
Q1 CHEMISTRY, MEDICINAL
引用次数: 0
Abstract
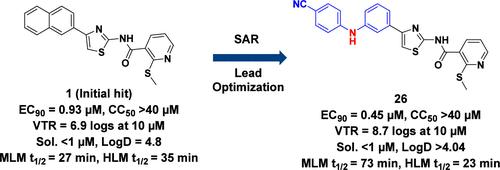
2-(Methylthio)-N-(4-(naphthalen-2-yl)thiazol-2-yl)nicotinamide 1 was identified as an inhibitor against Chikungunya virus (CHIKV) with good antiviral activity [EC50 = 0.6 μM; EC90 = 0.93 μM and viral titer reduction (VTR) of 6.9 logs at 10 μM concentration] with no observed cytotoxicity (CC50 = 132 μM) in normal human dermal fibroblast (NHDF) cells. Structure–activity relationship (SAR) studies to further improve the potency, efficacy, and drug-like properties of 1 led to the discovery of a new potent inhibitor N-(4-(3-((4-cyanophenyl)amino)phenyl)thiazol-2-yl)-2-(methylthio)nicotinamide 26, which showed a VTR of 8.7 logs at 10 μM against CHIKV and an EC90 of 0.45 μM with considerably improved MLM stability (t1/2 = 74 min) as compared to 1. Mechanism of action studies show that 26 inhibits alphavirus replication by blocking subgenomic viral RNA translation and structural protein synthesis. The in vivo efficacy studies of compound 26 on CHIKV infection in mice are reported.
求助全文
约1分钟内获得全文
求助全文
来源期刊

Journal of Medicinal Chemistry
医学-医药化学
CiteScore
4.00
自引率
11.00%
发文量
804
审稿时长
1.9 months
期刊介绍:
The Journal of Medicinal Chemistry is a prestigious biweekly peer-reviewed publication that focuses on the multifaceted field of medicinal chemistry. Since its inception in 1959 as the Journal of Medicinal and Pharmaceutical Chemistry, it has evolved to become a cornerstone in the dissemination of research findings related to the design, synthesis, and development of therapeutic agents.
The Journal of Medicinal Chemistry is recognized for its significant impact in the scientific community, as evidenced by its 2022 impact factor of 7.3. This metric reflects the journal's influence and the importance of its content in shaping the future of drug discovery and development. The journal serves as a vital resource for chemists, pharmacologists, and other researchers interested in the molecular mechanisms of drug action and the optimization of therapeutic compounds.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


