2′-Hydroxycinnamaldehyde induces ROS-mediated apoptosis in cancer cells by targeting PRX1 and PRX2
IF 4.2
Q2 CHEMISTRY, MULTIDISCIPLINARY
引用次数: 0
Abstract
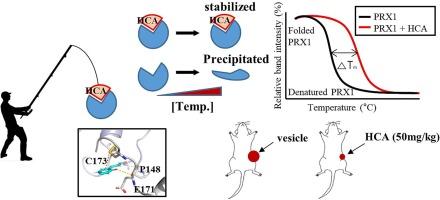
2′-Hydroxycinnamaldehyde (HCA) is a component of the commonly used spice cinnamon, which has beneficial effects on cancer, allergies, bacterial/viral infections, and Alzheimer’s disease. Our previous study showed that HCA induced reactive oxygen species (ROS) and apoptosis in cancer cells, and pretreatment of cancer cells with antioxidants abolished HCA-mediated ROS production and apoptosis. This indicates that ROS are critical effector for HCA activity. However, the molecular target of HCA for ROS induction has not been identified. In the present study, we identified peroxiredoxin 1 (PRX1) and peroxiredoxin 2 (PRX2) as target proteins of HCA using affinity chromatography, and further confirmed these association using a cellular thermal shift assay (CETSA). In addition, we used mutagenesis to identify important cysteine residues in PRX1 for HCA binding. PRX1 has four cysteines (Cys52, Cys71, Cys83, and Cys173), and when Cys173 (but not the other cysteine sites) was mutated to serine, it was unable to bind biotin-conjugated HCA, suggesting that Cys173 is important for HCA binding. Treatment of SW620 cancer cells transfected by control vector with 20 μM HCA increased ROS levels by 5.2-fold compared to DMSO-treated cells. However, downregulation of target proteins PRX1 and PRX2 using shRNAs (short hairpin RNA) significantly reduced HCA-mediated ROS induction (1.6-fold), supporting that PRX1 and PRX2 are targets of HCA for ROS elevation. Additionally, intraperitoneal injection of 50 mg/kg HCA inhibited SW620 tumor growth, resulting in a 59.9 % reduction in tumor volume. CETSA analysis of tumor tissues showed that PRX1 and PRX2 were bound and thus inactivated by HCA in a mouse xenograft model. These findings demonstrate that PRX1 and PRX2 are molecular target proteins responsible for HCA-induced ROS elevation and cancer cell death.
2′-羟基肉桂醛通过靶向PRX1和PRX2诱导ros介导的癌细胞凋亡
2 ' -羟基肉桂醛(HCA)是常用香料肉桂的一种成分,对癌症、过敏、细菌/病毒感染和阿尔茨海默病有有益作用。我们之前的研究表明,HCA诱导了癌细胞的活性氧(ROS)和凋亡,用抗氧化剂预处理癌细胞可以消除HCA介导的ROS的产生和凋亡。这表明活性氧是HCA活性的重要影响因子。然而,HCA诱导ROS的分子靶点尚未确定。在本研究中,我们通过亲和层析鉴定了过氧化氧还蛋白1 (PRX1)和过氧化氧还蛋白2 (PRX2)是HCA的靶蛋白,并通过细胞热移测定(CETSA)进一步证实了它们之间的关联。此外,我们使用诱变技术鉴定了PRX1中与HCA结合的重要半胱氨酸残基。PRX1有4种半胱氨酸(Cys52、Cys71、Cys83和Cys173),当Cys173(而不是其他半胱氨酸位点)突变为丝氨酸时,它不能结合生物素偶联的HCA,这表明Cys173对HCA的结合很重要。用对照载体转染20 μM HCA处理SW620癌细胞时,ROS水平较dmso处理细胞提高5.2倍。然而,使用shrna(短发夹RNA)下调靶蛋白PRX1和PRX2可显著降低HCA介导的ROS诱导(1.6倍),支持PRX1和PRX2是HCA升高ROS的靶点。此外,腹腔注射50 mg/kg HCA抑制SW620肿瘤生长,导致肿瘤体积减少59.9%。肿瘤组织的CETSA分析显示,在小鼠异种移植模型中,PRX1和PRX2被HCA结合并灭活。这些发现表明PRX1和PRX2是hca诱导的ROS升高和癌细胞死亡的分子靶蛋白。
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文

 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


