Design and synthesis of phenothiazine-based D–A molecules with ICT characteristics as efficient fluorescent probes for detecting hypochlorite in water
IF 4.3
2区 化学
Q1 SPECTROSCOPY
Spectrochimica Acta Part A: Molecular and Biomolecular Spectroscopy
Pub Date : 2024-11-26
DOI:10.1016/j.saa.2024.125491
引用次数: 0
Abstract
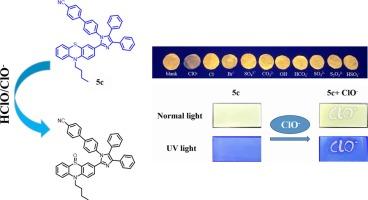
Phenylthiazide compounds are frequently utilized to construct electron donor–acceptor (D–A) dyes, exhibiting excellent fluorescence and colorimetric analysis performance due to their electron-rich properties. Three D-A type compounds 5a–c, featuring intramolecular charge transfer (ICT) characteristics, were designed and synthesized using phenothiazine as the donor and various aryl-substituted imidazoles as acceptors for the detection of hypochlorite. Additionally, the ICT properties of the compounds can be fine-tuned by introducing various substituents to achieve a more sensitive optical response to HClO/ClO−. Among these, compound 5c, which contains the electron-withdrawing group –CN, demonstrated high sensitivity (3.5 nM), high selectivity, and a rapid response to HClO/ClO−. Experimental results indicated that the strategy based on structural tuning presents a promising approach for optimizing fluorescent probes. To broaden practical applications, compound 5c was fabricated into paper test strips and thin-layer chromatography (TLC) plates for on-site analysis and detection of HClO/ClO−. The compound was capable of detecting HClO/ClO− not only in actual water samples but also in complex environmental water samples, thereby providing a convenient tool for real-time HClO/ClO− detection.
具有ICT特性的吩噻嗪基D-A分子的设计与合成及其在水中次氯酸盐检测中的高效荧光探针
苯噻嗪类化合物经常被用来构建电子给受体(D-A)染料,由于其富电子的性质,表现出优异的荧光和比色分析性能。以吩噻嗪为给体,各种芳基取代咪唑为受体,设计合成了3个具有分子内电荷转移(ICT)特征的D-A型化合物5a-c,用于次氯酸盐的检测。此外,化合物的ICT性质可以通过引入各种取代基来微调,以实现对HClO/ClO−更敏感的光学响应。其中,含有吸电子基团-CN的化合物5c对HClO/ClO−具有高灵敏度(3.5 nM)、高选择性和快速响应。实验结果表明,基于结构调谐的策略是一种很有前途的优化荧光探针的方法。为了扩大实际应用范围,将化合物5c制成纸质试纸条和薄层色谱(TLC)板,用于HClO/ClO−的现场分析和检测。该化合物不仅能检测实际水样中的HClO/ClO−,还能检测复杂环境水样中的HClO/ClO−,为实时检测HClO/ClO−提供了方便的工具。
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文
来源期刊
CiteScore
8.40
自引率
11.40%
发文量
1364
审稿时长
40 days
期刊介绍:
Spectrochimica Acta, Part A: Molecular and Biomolecular Spectroscopy (SAA) is an interdisciplinary journal which spans from basic to applied aspects of optical spectroscopy in chemistry, medicine, biology, and materials science.
The journal publishes original scientific papers that feature high-quality spectroscopic data and analysis. From the broad range of optical spectroscopies, the emphasis is on electronic, vibrational or rotational spectra of molecules, rather than on spectroscopy based on magnetic moments.
Criteria for publication in SAA are novelty, uniqueness, and outstanding quality. Routine applications of spectroscopic techniques and computational methods are not appropriate.
Topics of particular interest of Spectrochimica Acta Part A include, but are not limited to:
Spectroscopy and dynamics of bioanalytical, biomedical, environmental, and atmospheric sciences,
Novel experimental techniques or instrumentation for molecular spectroscopy,
Novel theoretical and computational methods,
Novel applications in photochemistry and photobiology,
Novel interpretational approaches as well as advances in data analysis based on electronic or vibrational spectroscopy.

 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


