Therapeutic landscape of metabolic dysfunction-associated steatohepatitis (MASH)
IF 122.7
1区 医学
Q1 BIOTECHNOLOGY & APPLIED MICROBIOLOGY
引用次数: 0
Abstract
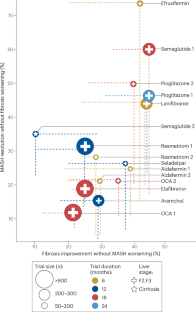
Metabolic dysfunction-associated steatotic liver disease (MASLD) and its severe subgroup metabolic dysfunction-associated steatohepatitis (MASH) have become a global epidemic and are driven by chronic overnutrition and multiple genetic susceptibility factors. The physiological outcomes include hepatocyte death, liver inflammation and cirrhosis. The first therapeutic for MASLD and MASH, resmetirom, has recently been approved for clinical use and has energized this therapeutic space. However, there is still much to learn in clinical studies of MASH, such as the scale of placebo responses, optimal trial end points, the time required for fibrosis reversal and side effect profiles. This Review introduces aspects of disease pathogenesis related to drug development and discusses two main therapeutic approaches. Thyroid hormone receptor-β agonists, such as resmetirom, as well as fatty acid synthase inhibitors, target the liver and enable it to function within a toxic metabolic environment. In parallel, incretin analogues such as semaglutide improve metabolism, allowing the liver to self-regulate and reversing many aspects of MASH. We also discuss how combinations of therapeutics could potentially be used to treat patients. Driven by chronic overnutrition and associated with obesity, metabolic dysfunction-associated steatotic liver disease is increasing in prevalence. This Review discusses two main therapeutic approaches: improving the metabolism with incretin mimetics such as semaglutide and directly targeting the liver with agents such as the recently approved resmetirom.

代谢功能障碍相关性脂肪性肝炎(MASH)的治疗前景
代谢功能障碍相关性脂肪性肝病(MASLD)及其严重亚组代谢功能障碍相关性脂肪性肝炎(MASH)已成为一种全球性流行病,其成因是长期营养过剩和多种遗传易感因素。其生理结果包括肝细胞死亡、肝脏炎症和肝硬化。治疗 MASLD 和 MASH 的第一种疗法 resmetirom 最近已被批准用于临床,为这一治疗领域注入了活力。然而,在MASH的临床研究中仍有许多东西需要学习,如安慰剂反应的规模、最佳试验终点、纤维化逆转所需的时间和副作用特征等。本综述介绍了与药物开发相关的疾病发病机制,并讨论了两种主要的治疗方法。甲状腺激素受体-β激动剂(如雷美替罗)和脂肪酸合成酶抑制剂以肝脏为靶点,使其在有毒代谢环境中发挥作用。与此同时,胰高血糖素类似物(如semaglutide)可改善新陈代谢,使肝脏能够自我调节并逆转MASH的许多方面。我们还讨论了如何将各种疗法结合起来治疗患者。
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文
来源期刊

Nature Reviews. Drug Discovery
医学-生物工程与应用微生物
CiteScore
137.40
自引率
0.30%
发文量
227
期刊介绍:
Nature Reviews Drug Discovery is a monthly journal aimed at everyone working in the drug discovery and development arena.
Each issue includes:
Highest-quality reviews and perspectives covering a broad scope.
News stories investigating the hottest topics in drug discovery.
Timely summaries of key primary research papers.
Concise updates on the latest advances in areas such as new drug approvals, patent law, and emerging industry trends and strategies.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


