Arabinogalactan from Cynanchum atratum induces tolerogenic dendritic cells in gut to restrain autoimmune response and alleviate collagen-induced arthritis in mice
IF 6.7
1区 医学
Q1 CHEMISTRY, MEDICINAL
引用次数: 0
Abstract
Background
Rheumatoid arthritis (RA) is an autoimmune disease characterized by multiple joints lesions. Tolerogenic dendritic cells (tolDCs) play crucial roles in maintaining immune homeostasis. The immunomodulatory activity of plant-derived arabinogalactan (AGs) has been well investigated, however, whether AGs could suppress autoimmune responses by inducing tolDCs is remain unclear.
Design
Collagen-induced arthritis (CIA, a mouse model of RA) mice were utilized to ascertain the role of AGs (obtained from Cynanchum atratum) in autoimmune responses. An antibiotic cocktail was administered to eliminate gut microbiota. Germ-free (GF) and Toll-like receptor 2 (TLR2) knockout mice were used to determine the function of AGs in intestinal immune cells.
Results
The oral administration of dietary AGs substantially reduced the severity of CIA and rebalanced the ratio of regulatory T cells (Tregs) to T helper 17 (Th17) cells. Although the antibiotic cocktail depleted the mice's gut microbiota, AGs had a therapeutic effect on their CIA. AGs restored Treg/Th17 homeostasis by inducing CD103+ tolDCs, regardless of the gut microbiota of the GF mice. Coculture experiments confirmed that AGs induced tolDCs and transforming growth factor β (TGF-β) secretion, leading to Treg amplification. RNA sequencing and TLR2 knockout experiments revealed that AGs induced tolDCs through a TLR2-mediated mechanism. Preventive interventions with AGs established a tolerogenic intestinal immune microenvironment, which delayed the onset and progression of CIA. AGs functioned synergistically with tofacitinib, a JAK inhibitor, to effectively restore Treg/Th17 balance and alleviate CIA.
Conclusion
This study introduces a novel microbiota-independent mechanism through which soluble dietary AGs inhibit systemic autoimmune responses. Our findings provide insights into the supplementation of dietary AGs in patients with preclinical or progressive RA.
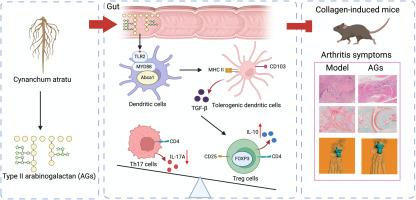
蝙蝠葛中的阿拉伯半乳聚糖诱导肠道中的耐受性树突状细胞,从而抑制自身免疫反应并缓解胶原蛋白诱导的小鼠关节炎
背景类风湿性关节炎(RA)是一种以多发性关节病变为特征的自身免疫性疾病。耐受性树突状细胞(tolerogenic dendritic cells,tolDCs)在维持免疫平衡方面发挥着至关重要的作用。设计利用胶原诱导的关节炎(CIA,一种RA的小鼠模型)小鼠来确定AGs(取自Cynanchum atratum)在自身免疫反应中的作用。给小鼠注射抗生素鸡尾酒以消除肠道微生物群。结果口服膳食AGs大大降低了CIA的严重程度,并重新平衡了调节性T细胞(Tregs)与T辅助细胞17(Th17)的比例。虽然抗生素鸡尾酒耗尽了小鼠的肠道微生物群,但AGs对小鼠的CIA有治疗作用。AGs 通过诱导 CD103+ tolDCs 恢复了 Treg/Th17 的平衡,而与 GF 小鼠的肠道微生物群无关。共培养实验证实,AGs能诱导tolDCs和转化生长因子β(TGF-β)分泌,从而导致Treg扩增。RNA测序和TLR2基因敲除实验显示,AGs通过TLR2介导的机制诱导tolDCs。用AGs进行预防性干预可建立一种耐受性肠道免疫微环境,从而延缓CIA的发病和进展。AGs与JAK抑制剂托法替尼协同作用,可有效恢复Treg/Th17平衡并缓解CIA。我们的研究结果为临床前或进展期 RA 患者补充膳食 AGs 提供了启示。
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文
来源期刊

Phytomedicine
医学-药学
CiteScore
10.30
自引率
5.10%
发文量
670
审稿时长
91 days
期刊介绍:
Phytomedicine is a therapy-oriented journal that publishes innovative studies on the efficacy, safety, quality, and mechanisms of action of specified plant extracts, phytopharmaceuticals, and their isolated constituents. This includes clinical, pharmacological, pharmacokinetic, and toxicological studies of herbal medicinal products, preparations, and purified compounds with defined and consistent quality, ensuring reproducible pharmacological activity. Founded in 1994, Phytomedicine aims to focus and stimulate research in this field and establish internationally accepted scientific standards for pharmacological studies, proof of clinical efficacy, and safety of phytomedicines.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


