Nrf2 activator tertiary butylhydroquinone enhances neural stem cell differentiation and implantation in Alzheimer’s disease by boosting mitochondrial function
IF 2.7
4区 医学
Q3 NEUROSCIENCES
引用次数: 0
Abstract
Aims
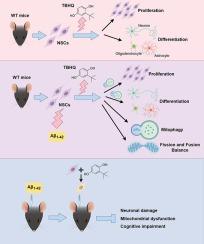
To investigate the effects of Nrf2 agonist tertiary butylhydroquinone (TBHQ)-stimulated neural stem cells (NSCs) transplantation (NSC(TBHQ)) on neuronal damage and cognitive deficits in an AD model and its underlying principles.
Methods
BHQ-treated NSCs were examined with or without Aβ1-42 to investigate the effects of TBHQ on the proliferation and differentiation functions. The mitophagy inhibitor Cyclosporine A (CSA) was used to explore the regulation of mitophagy by TBHQ. The no-, ethanol-, and TBHQ-treated NSCs were transplanted into the bilateral hippocampal region of model mice to explore the effects of NSC(TBHQ) on neuronal, cognitive, and mitochondrial functional impairments in mice.
Results
TBHQ reversed the Aβ1-42-caused inhibition on NSC proliferation and differentiation, as well as on levels of mitochondrial membrane potential, adenosine triphosphate (ATP), and mitochondrial fusion-associated proteins. TBHQ alleviated the Aβ1-42-induced increase in apoptosis, mitochondrial damage, mitochondria-derived reactive oxygen species (mtROS), and mitochondrial fission-related proteins. TBHQ activated the Parkin, Beclin, LC3II/I, and COXIV expression, while inhibiting the p62 expression. CSA reversed the effects of TBHQ on NSC proliferation and differentiation. After NSC(TBHQ) transplantation, it not only further extended the dwell time in the target quadrant and shorten the time and distance for finding the hidden platform, but also further decreased the Aβ and p-Tau/Tau levels, while increasing the expression of NeuN. The effects of NSC(TBHQ) transplantation on mitochondrial function were consistent with the in vitro experiments.
Conclusions
The study shows that NSC(TBHQ) intensifies the beneficial impact of NSCs transplantation on cognitive impairment and neuronal damage in AD models, likely due to TBHQ’s role in promoting NSCs growth and differentiation via mitophagy, thus laying a theoretical foundation for improving NSCs transplantation for AD.
Nrf2激活剂叔丁基对苯二酚通过增强线粒体功能提高阿尔茨海默病神经干细胞的分化和植入。
目的:研究Nrf2激动剂叔丁基对苯二酚(TBHQ)刺激神经干细胞(NSCs)移植(NSC(TBHQ))对AD模型神经元损伤和认知障碍的影响及其基本原理:方法:在有或无Aβ1-42的情况下检测BHQ处理的NSCs,以研究TBHQ对其增殖和分化功能的影响。有丝分裂抑制剂环孢菌素 A(CSA)被用来探索 TBHQ 对有丝分裂的调控。将未经处理、乙醇处理和TBHQ处理的NSCs移植到模型小鼠的双侧海马区,探讨NSC(TBHQ)对小鼠神经元、认知和线粒体功能损伤的影响:结果:TBHQ逆转了Aβ1-42对NSC增殖和分化的抑制,以及对线粒体膜电位、三磷酸腺苷(ATP)和线粒体融合相关蛋白水平的抑制。TBHQ 可减轻 Aβ1-42- 诱导的细胞凋亡、线粒体损伤、线粒体衍生活性氧(mtROS)和线粒体裂变相关蛋白的增加。TBHQ 激活了 Parkin、Beclin、LC3II/I 和 COXIV 的表达,同时抑制了 p62 的表达。CSA 逆转了 TBHQ 对 NSC 增殖和分化的影响。NSC(TBHQ)移植后,不仅进一步延长了在目标象限的停留时间,缩短了寻找隐藏平台的时间和距离,还进一步降低了Aβ和p-Tau/Tau的水平,同时增加了NeuN的表达。NSC(TBHQ)移植对线粒体功能的影响与体外实验结果一致:研究表明,NSC(TBHQ)能增强NSCs移植对AD模型认知障碍和神经元损伤的有益影响,这可能是由于TBHQ能通过有丝分裂促进NSCs的生长和分化,从而为改善NSCs移植治疗AD奠定了理论基础。
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文
来源期刊

Brain Research
医学-神经科学
CiteScore
5.90
自引率
3.40%
发文量
268
审稿时长
47 days
期刊介绍:
An international multidisciplinary journal devoted to fundamental research in the brain sciences.
Brain Research publishes papers reporting interdisciplinary investigations of nervous system structure and function that are of general interest to the international community of neuroscientists. As is evident from the journals name, its scope is broad, ranging from cellular and molecular studies through systems neuroscience, cognition and disease. Invited reviews are also published; suggestions for and inquiries about potential reviews are welcomed.
With the appearance of the final issue of the 2011 subscription, Vol. 67/1-2 (24 June 2011), Brain Research Reviews has ceased publication as a distinct journal separate from Brain Research. Review articles accepted for Brain Research are now published in that journal.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


