Engineered multienzyme-mimicking 2D bismuthene catalytic nanotriggers enable cascade enzyodynamic-boosted and synergistic GPX4/FSP1-mediated ferroptosis amplification for cancer radiosensitization
IF 13.3
1区 工程技术
Q1 ENGINEERING, CHEMICAL
引用次数: 0
Abstract
Radiotherapy (RT) is a critical clinical treatment for cancer. However, radioresistance often hampers its effectiveness, leading to local recurrence and therapeutic failure. Ferroptosis has been regarded as a natural barrier to tumor progression and plays a significant role in RT-mediated anticancer effects. Therefore, the simultaneous activation of ferroptosis and RT is of great significance for cancer therapy. Herein, we engineered the tumor-releasing nanozymes (BMBs), combining manganese oxide as the ferroptosis inducer and two-dimensional bismuthene with high-Z effect for augmented ferroptotic RT in a triple-enzyme-like radiosensitization manner. The nanozymes BMBs depleted glutathione (GSH) and nicotinamide adenine dinucleotide phosphate (NADPH) in the tumor microenvironment to exhibit glutathione peroxidase (GPX)-like activity and NADPH dehydrogenase (NDH)-like activity, accompanied by aberrant reactive oxygen species (ROS) production exhibiting peoxidase (POD)-like activity. In addition, the nanozymes BMBs simultaneously inactivate ferroptosis defensive system: glutathione peroxidase 4 (GPX4) and ferroptosis suppressor protein 1 (FSP1) to induce ferroptosis. The strong oxidative stress induced cascade enzyodynamic effect and ferroptosis, which synergized with the two-dimensional bismuthene-mediated radiosensitization to improve the efficacy of RT. Both in vitro and in vivo experiments substantiated the excellent radiotherapeutic response of the nanozymes by enhancing RT and ferroptosis. Therefore, this work demonstrates that the rational combination of nanozymes with POD/GPX/NDH-like activity and GPX4/FSP1 suppressing ability to induce ferroptosis for synergistic radiosensitization provides a viable and promising strategy for cancer treatment.
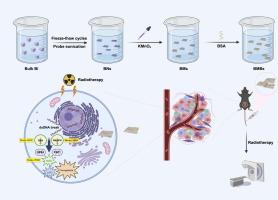
工程化多酶模拟二维双钌催化纳米触发器可实现级联酵素动力促进和协同 GPX4/FSP1 介导的铁氧化酶放大作用,从而实现癌症放射增敏
放疗(RT)是治疗癌症的重要临床手段。然而,放射抗药性往往会阻碍其疗效,导致局部复发和治疗失败。铁突变被认为是肿瘤进展的天然屏障,在 RT 介导的抗癌效应中发挥着重要作用。因此,同时激活铁突变和 RT 对癌症治疗具有重要意义。在此,我们设计了肿瘤释放纳米酶(BMBs),将氧化锰作为铁突变诱导剂与具有高Z效应的二维双钌相结合,以三酶样放射增敏方式增强铁突变RT。纳米酶 BMBs 消耗了肿瘤微环境中的谷胱甘肽(GSH)和烟酰胺腺嘌呤二核苷酸磷酸酯(NADPH),表现出类似谷胱甘肽过氧化物酶(GPX)的活性和类似 NADPH 脱氢酶(NDH)的活性,并伴随着异常活性氧(ROS)生成,表现出类似 peoxidase(POD)的活性。此外,纳米酶 BMBs 还同时使谷胱甘肽过氧化物酶 4 (GPX4) 和铁氧化抑制蛋白 1 (FSP1) 这两个铁氧化防御系统失活,从而诱导铁氧化。强氧化应激诱导了级联酶动力效应和铁突变,与二维双钌介导的放射增敏协同作用,提高了 RT 的疗效。体外和体内实验都证实了纳米酶通过增强 RT 和铁跃迁而产生的良好放射治疗反应。因此,这项工作表明,将具有 POD/GPX/NDH 类活性和 GPX4/FSP1 抑制能力的纳米酶合理地结合在一起,诱导铁变态反应,从而实现协同放射增敏,为癌症治疗提供了一种可行且前景广阔的策略。
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文
来源期刊

Chemical Engineering Journal
工程技术-工程:化工
CiteScore
21.70
自引率
9.30%
发文量
6781
审稿时长
2.4 months
期刊介绍:
The Chemical Engineering Journal is an international research journal that invites contributions of original and novel fundamental research. It aims to provide an international platform for presenting original fundamental research, interpretative reviews, and discussions on new developments in chemical engineering. The journal welcomes papers that describe novel theory and its practical application, as well as those that demonstrate the transfer of techniques from other disciplines. It also welcomes reports on carefully conducted experimental work that is soundly interpreted. The main focus of the journal is on original and rigorous research results that have broad significance. The Catalysis section within the Chemical Engineering Journal focuses specifically on Experimental and Theoretical studies in the fields of heterogeneous catalysis, molecular catalysis, and biocatalysis. These studies have industrial impact on various sectors such as chemicals, energy, materials, foods, healthcare, and environmental protection.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


