Human genetics identify convergent signals in mitochondrial LACTB-mediated lipid metabolism in cardiovascular-kidney-metabolic syndrome
IF 27.7
1区 生物学
Q1 CELL BIOLOGY
引用次数: 0
Abstract
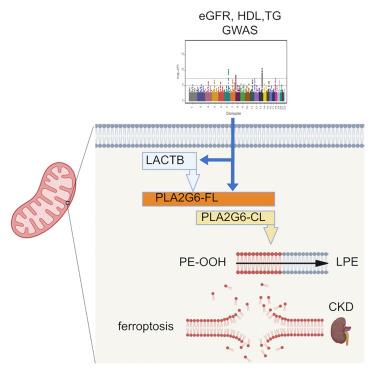
The understanding of cardiovascular-kidney-metabolic syndrome remains difficult despite recently performed large scale genome-wide association studies. Here, we identified beta-lactamase (LACTB), a novel gene whose expression is targeted by genetic variations causing kidney dysfunction and hyperlipidemia. Mice with LACTB deletion developed impaired glucose tolerance, elevated lipid levels, and increased sensitivity to kidney disease, while mice with tubule-specific overexpression of LACTB were protected from kidney injury. We show that LACTB is a novel mitochondrial protease cleaving and activating phospholipase A2 group VI (PLA2G6), a kidney-metabolic risk gene itself. Genetic deletion of PLA2G6 in tubule-specific LACTB-overexpressing mice abolished the protective function of LACTB. Via mouse and human lipidomic studies, we show that LACTB and downstream PLA2G6 convert oxidized phosphatidylethanolamine to lyso-phosphatidylethanolamine and thereby regulate mitochondrial function and ferroptosis. In summary, we identify a novel gene and a core targetable pathway for kidney-metabolic disorders.
人类遗传学发现心血管-肾脏-代谢综合征线粒体 LACTB 介导的脂质代谢中存在趋同信号
尽管最近进行了大规模的全基因组关联研究,但人们对心血管-肾脏-代谢综合征的了解仍然十分困难。在这里,我们发现了β-内酰胺酶(LACTB),这是一种新型基因,其表达受导致肾功能障碍和高脂血症的基因变异的影响。缺失 LACTB 的小鼠会出现糖耐量受损、血脂水平升高以及对肾脏疾病的敏感性增加,而肾小管特异性过表达 LACTB 的小鼠则可免受肾脏损伤。我们发现,LACTB 是一种新型线粒体蛋白酶,能裂解和激活磷脂酶 A2 组 VI(PLA2G6),而磷脂酶 A2 组 VI 本身就是一种肾脏代谢风险基因。在肾小管特异性 LACTB 基因表达过高的小鼠体内遗传性地删除 PLA2G6,会取消 LACTB 的保护功能。通过小鼠和人类脂质体研究,我们发现 LACTB 及其下游的 PLA2G6 能将氧化磷脂酰乙醇胺转化为溶血磷脂酰乙醇胺,从而调节线粒体功能和铁蛋白沉积。总之,我们发现了一个新基因和肾脏代谢紊乱的核心靶向途径。
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文
来源期刊
Cell metabolism
生物-内分泌学与代谢
CiteScore
48.60
自引率
1.40%
发文量
173
审稿时长
2.5 months
期刊介绍:
Cell Metabolism is a top research journal established in 2005 that focuses on publishing original and impactful papers in the field of metabolic research.It covers a wide range of topics including diabetes, obesity, cardiovascular biology, aging and stress responses, circadian biology, and many others.
Cell Metabolism aims to contribute to the advancement of metabolic research by providing a platform for the publication and dissemination of high-quality research and thought-provoking articles.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


