Discovery of 4-Hydroxyphenylpyruvate dioxygenase inhibitors with novel pharmacophores
引用次数: 0
Abstract
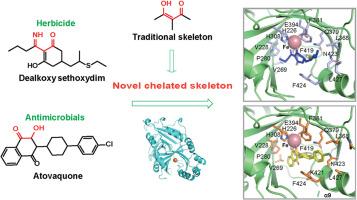
4-Hydroxyphenylpyruvate dioxygenase (HPPD) is pivotal in tyrosine metabolism and essential for plant survival. Its inhibition leads to leaf bleaching and plant death. While current HPPD inhibitors are effective, they pose phytotoxicity risks and may contribute to herbicide resistance. Here, we investigated the inhibitory potential of sethoxydim and atovaquone, which traditionally target acetyl-CoA carboxylase and the cytochrome bc1 complex, respectively. Both atovaquone and the degradation product of sethoxydim exhibited moderate HPPD inhibitory activity. But the mechanism by which sethoxydim inhibited HPPD remained unclear. Therefore, we embarked on an investigation into the crystal structure of the complex, with the aim of elucidating its precise binding mode. Our findings revealed that sethoxydim degrades in solution, producing dealkoxy sethoxydim as the active component in HPPD inhibition. Structural analysis elucidated the binding modes of atovaquone and dealkoxy sethoxydim with HPPD. These binding motifs represent novel pharmacophores and offer promising leads for developing HPPD inhibitors with improved pesticidal profiles.
发现具有新药理作用的 4-羟基苯基丙酮酸二氧酶抑制剂
4-Hydroxyphenylpyruvate dioxygenase(HPPD)在酪氨酸代谢中起着关键作用,对植物的生存至关重要。抑制该酶会导致叶片脱色和植物死亡。虽然目前的 HPPD 抑制剂很有效,但它们存在植物毒性风险,并可能导致除草剂抗药性。在这里,我们研究了sethoxydim 和阿托伐醌的抑制潜力,它们传统上分别以乙酰-CoA 羧化酶和细胞色素 bc1 复合物为靶标。阿托伐醌和sethoxydim 的降解产物都表现出中等程度的 HPPD 抑制活性。但sethoxydim抑制HPPD的机制仍不清楚。因此,我们开始研究该复合物的晶体结构,旨在阐明其精确的结合模式。我们的研究结果表明,sethoxydim 在溶液中降解,产生的脱烷氧基 sethoxydim 是抑制 HPPD 的活性成分。结构分析阐明了阿托伐醌和脱烷氧基sethoxydim与HPPD的结合模式。这些结合基团代表了新的药性,为开发具有更好杀虫特性的 HPPD 抑制剂提供了很好的线索。
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文

 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


