Innovative spiral nerve conduits: Addressing nutrient transport and cellular activity for critical-sized nerve defects
IF 18
1区 医学
Q1 ENGINEERING, BIOMEDICAL
引用次数: 0
Abstract
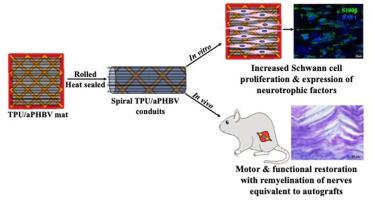
Large-gap nerve defects require nerve guide conduits (NGCs) for complete regeneration and muscle innervation. Many NGCs have been developed using various scaffold designs and tissue engineering strategies to promote axon regeneration. Still, most are tubular with inadequate pore sizes and lack surface cues for nutrient transport, cell attachment, and tissue infiltration. This study developed a porous spiral NGC to address these issues using a 3D-printed thermoplastic polyurethane (TPU) fiber lattice. The lattice was functionalized with poly(3-hydroxybutyrate-co-3-hydroxyvalerate) (PHBV) electrospun aligned (aPHBV) and randomly (rPHBV) oriented nanofibers to enhance cellular activity. TPU lattices were made with 25 %, 35 %, and 50 % infill densities to create scaffolds with varied mechanical compliance. The fabricated TPU/PHBV spiral conduits had significantly higher surface areas (25 % TPU/PHBV: 698.97 mm2, 35 % TPU/PHBV: 500.06 mm2, 50 % TPU/PHBV: 327.61 mm2) compared to commercially available nerve conduits like Neurolac™ (205.26 mm2). Aligned PHBV nanofibers showed excellent Schwann cell (RSC96) adhesion, proliferation, and neurogenic gene expression for all infill densities. Spiral TPU/PHBV conduits with 25 % and 35 % infill densities exhibited Young's modulus values comparable to Neurotube® and ultimate tensile strength like acellular cadaveric human nerves. A 10 mm sciatic nerve defect in Wistar rats treated with TPU/aPHBV NGCs demonstrated muscle innervation and axon healing comparable to autografts over 4 months, as evaluated by gait analysis, functional recovery, and histology. The TPU/PHBV NGC developed in this study shows promise as a treatment for large-gap nerve defects.
创新性螺旋神经导管:解决临界尺寸神经缺损的营养运输和细胞活动问题
大间隙神经缺损需要神经引导导管(NGC)来实现完全再生和肌肉神经支配。为了促进轴突再生,人们利用各种支架设计和组织工程策略开发了许多 NGC。然而,大多数NGC都是管状的,孔隙大小不足,缺乏营养运输、细胞附着和组织浸润的表面线索。本研究利用 3D 打印热塑性聚氨酯(TPU)纤维晶格开发了一种多孔螺旋 NGC,以解决这些问题。为了增强细胞活性,该晶格用聚合(3-羟基丁酸-co-3-羟基戊酸)(PHBV)电纺排列(aPHBV)和随机(rPHBV)取向纳米纤维进行了功能化。TPU 网格的填充密度分别为 25%、35% 和 50%,以创建具有不同机械顺应性的支架。与 Neurolac™ 等市售神经导管(205.26 平方毫米)相比,制成的热塑性聚氨酯/PHBV 螺旋导管的表面积明显更高(25% 热塑性聚氨酯/PHBV:698.97 平方毫米;35% 热塑性聚氨酯/PHBV:500.06 平方毫米;50% 热塑性聚氨酯/PHBV:327.61 平方毫米)。排列整齐的 PHBV 纳米纤维在所有填充密度下都显示出出色的许旺细胞(RSC96)粘附、增殖和神经源基因表达能力。填充密度分别为 25% 和 35% 的螺旋形热塑性聚氨酯/PHBV 导管的杨氏模量值与 Neurotube® 不相上下,极限拉伸强度与无细胞尸体人类神经相当。通过步态分析、功能恢复和组织学评估,使用 TPU/aPHBV NGCs 治疗 Wistar 大鼠 10 毫米坐骨神经缺损 4 个月后,其肌肉神经支配和轴突愈合情况与自体移植相当。本研究中开发的 TPU/PHBV NGC 具有治疗大间隙神经缺损的前景。
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文
来源期刊

Bioactive Materials
Biochemistry, Genetics and Molecular Biology-Biotechnology
CiteScore
28.00
自引率
6.30%
发文量
436
审稿时长
20 days
期刊介绍:
Bioactive Materials is a peer-reviewed research publication that focuses on advancements in bioactive materials. The journal accepts research papers, reviews, and rapid communications in the field of next-generation biomaterials that interact with cells, tissues, and organs in various living organisms.
The primary goal of Bioactive Materials is to promote the science and engineering of biomaterials that exhibit adaptiveness to the biological environment. These materials are specifically designed to stimulate or direct appropriate cell and tissue responses or regulate interactions with microorganisms.
The journal covers a wide range of bioactive materials, including those that are engineered or designed in terms of their physical form (e.g. particulate, fiber), topology (e.g. porosity, surface roughness), or dimensions (ranging from macro to nano-scales). Contributions are sought from the following categories of bioactive materials:
Bioactive metals and alloys
Bioactive inorganics: ceramics, glasses, and carbon-based materials
Bioactive polymers and gels
Bioactive materials derived from natural sources
Bioactive composites
These materials find applications in human and veterinary medicine, such as implants, tissue engineering scaffolds, cell/drug/gene carriers, as well as imaging and sensing devices.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


