Hyaluronic acid-functionalized ruthenium photothermal nanoenzyme for enhancing osteosarcoma chemotherapy: Cascade targeting and bidirectional modulation of drug resistance
IF 10.7
1区 化学
Q1 CHEMISTRY, APPLIED
引用次数: 0
Abstract
Insufficient drug delivery efficiency in vivo and robust drug resistance are two major factors to induce suboptimal efficacy in chemotherapy of osteosarcoma (OS). To address these challenges, we developed polysaccharide hyaluronic acid (HA)-functionalized ruthenium nanoaggregates (Ru NAs) to enhance the chemotherapy of doxorubicin (DOX) for OS. These NAs, comprising Ru nanoparticles (NPs) and alendronate-modified HA (HA-ALN), effectively load DOX, resulting in DOX@Ru-HA-ALN NAs. The combination of HA and ALN in NAs ensures outstanding cascade targeting towards tumor-invaded bone tissues and CD44-overexpressing tumor cells, maximizing therapeutic efficacy while minimizing off-target effects. Concurrently, the Ru NPs in NAs function as “smart” photoenzymatic agent to not only in situ relieve hypoxia of OS via the catalysis of overexpressed H2O2 to produce O2, but also generate mild photothermal effect under 808-nm laser irradiation. They can bidirectionally overcome drug resistance of DOX via downregulation of resistance-related factors including multi-drug resistant associate protein, P-glycoprotein, heat shock factor 1, etc. The integration of cascade targeting with bidirectional modulation of drug resistance positions Ru-HA-ALN NAs to substantially enhance DOX chemotherapy for OS. Therefore, the present work highlights the potential of polysaccharide-functionalized nanomaterials in advancing tumor chemotherapy by addressing challenges of both delivery efficiency and drug resistance.
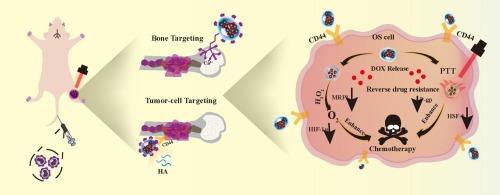
用于增强骨肉瘤化疗的透明质酸功能化钌光热纳米酶:级联靶向和双向调节耐药性
体内给药效率低和耐药性强是导致骨肉瘤(OS)化疗疗效不理想的两个主要因素。为了应对这些挑战,我们开发了多糖透明质酸(HA)功能化的钌纳米聚集体(Ru NAs),以增强多柔比星(DOX)对骨肉瘤的化疗效果。这些由 Ru 纳米颗粒(NPs)和阿仑膦酸修饰的 HA(HA-ALN)组成的 NAs 能有效负载 DOX,形成 DOX@Ru-HA-ALN NAs。NAs中HA和ALN的结合确保了对肿瘤侵袭的骨组织和CD44表达过高的肿瘤细胞的出色级联靶向性,在最大限度地提高疗效的同时减少了脱靶效应。同时,NAs 中的 Ru NPs 具有 "智能 "光酶制剂的功能,不仅能通过催化过表达的 H2O2 产生 O2 来缓解 OS 的原位缺氧,还能在 808 纳米激光照射下产生温和的光热效应。它们可以通过下调耐药相关因子(包括多重耐药联系蛋白、P-糖蛋白、热休克因子 1 等)双向克服 DOX 的耐药性。Ru-HA-ALN NAs将级联靶向与双向调节耐药性相结合,可大大提高OS的DOX化疗效果。因此,本研究强调了多糖功能化纳米材料在推进肿瘤化疗方面的潜力,解决了递送效率和耐药性两方面的难题。
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文
来源期刊

Carbohydrate Polymers
化学-高分子科学
CiteScore
22.40
自引率
8.00%
发文量
1286
审稿时长
47 days
期刊介绍:
Carbohydrate Polymers stands as a prominent journal in the glycoscience field, dedicated to exploring and harnessing the potential of polysaccharides with applications spanning bioenergy, bioplastics, biomaterials, biorefining, chemistry, drug delivery, food, health, nanotechnology, packaging, paper, pharmaceuticals, medicine, oil recovery, textiles, tissue engineering, wood, and various aspects of glycoscience.
The journal emphasizes the central role of well-characterized carbohydrate polymers, highlighting their significance as the primary focus rather than a peripheral topic. Each paper must prominently feature at least one named carbohydrate polymer, evident in both citation and title, with a commitment to innovative research that advances scientific knowledge.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


