Impact of protein aggregation on the immunogenicity of a human monoclonal antibody following pulmonary administration in mice
IF 5.3
2区 医学
Q1 PHARMACOLOGY & PHARMACY
引用次数: 0
Abstract
Spray drying is a widely employed method for generating dry powder formulations for inhalation. Yet, it presents substantial challenges when applied to therapeutic proteins due to stability issues. The formation of protein aggregates during the atomization and the heating steps can diminish protein activity and raise immunogenicity concerns. Here, we assessed the impact of varying levels of protein aggregates generated during spray-drying on the fate and the immunogenicity of the human monoclonal antibody NIP228 following intratracheal administration in mice. Aggregate-free rhodamine labelled NIP228 was spray-dried with or without 1% polysorbate 80 surfactant, resulting in the generation of powder formulations with associated low and high protein aggregate levels, respectively. Confocal imaging highlighted the presence of aggregates in the lungs for both powders but not for the solution following a single dose administration. Flow cytometry analysis designated alveolar macrophages as the main immune cells taking up rhod-NIP228 in the lungs with very little involvement of dendritic cells and interstitial macrophages. Notably, repeated intratracheal administration of the three formulations in mice did not impact the magnitude of the anti-drug antibody response in sera or broncho-alveolar lavages. Furthermore, the pulmonary route appeared to evoke a more robust immune response when compared to subcutaneous administration. Overall, the level of NIP228 aggregation in this study did not appear to be the primary driver of NIP228 immunogenicity following delivery to the lungs in mice.These findings shed new light on the interplay between protein aggregation and immunogenicity in the context of the pulmonary delivery of therapeutic proteins.
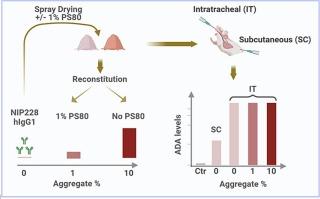
小鼠肺部给药后蛋白质聚集对人类单克隆抗体免疫原性的影响。
喷雾干燥法是一种广泛应用于生产吸入用干粉制剂的方法。然而,由于稳定性问题,这种方法在应用于治疗性蛋白质时面临巨大挑战。在雾化和加热步骤中形成的蛋白质聚集体会降低蛋白质的活性,并引发免疫原性问题。在此,我们评估了喷雾干燥过程中产生的不同程度的蛋白质聚集体对小鼠气管内给药后人类单克隆抗体 NIP228 的转归和免疫原性的影响。在喷雾干燥过程中添加或不添加 1%的聚山梨醇酯 80 表面活性剂,生成的粉末制剂分别具有相关的低蛋白聚集水平和高蛋白聚集水平。共焦成像显示,两种粉末在肺部都存在聚集体,而溶液则没有。流式细胞术分析表明,单剂量给药后,肺泡巨噬细胞是肺部吸收 rhod-NIP228 的主要免疫细胞,树突状细胞和间质巨噬细胞很少参与。值得注意的是,对小鼠反复气管内注射三种制剂不会影响血清或支气管肺泡灌洗液中抗药抗体反应的强度。与皮下注射相比,肺部途径似乎能唤起更强的免疫反应。总之,在这项研究中,NIP228的聚集程度似乎并不是NIP228在小鼠肺部给药后产生免疫原性的主要驱动因素,从而为治疗蛋白肺部给药过程中蛋白质聚集与免疫原性之间的相互作用提供了新的思路。
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文
来源期刊
CiteScore
10.70
自引率
8.60%
发文量
951
审稿时长
72 days
期刊介绍:
The International Journal of Pharmaceutics is the third most cited journal in the "Pharmacy & Pharmacology" category out of 366 journals, being the true home for pharmaceutical scientists concerned with the physical, chemical and biological properties of devices and delivery systems for drugs, vaccines and biologicals, including their design, manufacture and evaluation. This includes evaluation of the properties of drugs, excipients such as surfactants and polymers and novel materials. The journal has special sections on pharmaceutical nanotechnology and personalized medicines, and publishes research papers, reviews, commentaries and letters to the editor as well as special issues.

 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


