Pan-Cancer Single-Cell Transcriptomic Analysis Reveals Divergent Expression of Embryonic Proangiogenesis Gene Modules in Tumorigenesis
Abstract
Background
Angiogenesis is indispensable for the sustained survival and progression of both embryonic development and tumorigenesis. This intricate process is tightly regulated by a multitude of pro-angiogenic genes. The presence of gene modules facilitating angiogenesis has been substantiated in both embryonic development and the context of tumor proliferation. However, it remains unresolved whether the pro-angiogenic gene modules expressed during embryonic development also exist in tumors.
Methods
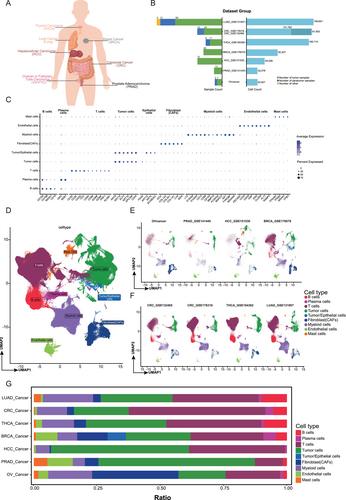
This study performed a pan-cancer single-cell RNA sequencing (scRNA-seq) analysis on samples from 332 patients across seven cancer types: thyroid carcinoma, lung cancer, breast cancer, hepatocellular carcinoma, colorectal cancer, ovarian carcinoma, and prostate adenocarcinoma. Data processing was carried out using the Seurat R package, with rigorous quality control to filter high-quality cells and mitigate batch effects across datasets. We used principal component analysis (PCA), shared nearest neighbor graph-based clustering, and Uniform Manifold Approximation and Projection (UMAP) to visualize cell types and identify distinct cell clusters. Myeloid cell subpopulations were further analyzed for the expression of embryonic pro-angiogenic gene modules (EPGM) and tumor pro-angiogenic gene modules (TPGM).
Results
The analysis identified nine major cell types within the tumor microenvironment, with myeloid cells consistently exhibiting elevated expression of both tumor pro-angiogenic gene modules (TPGM) and EPGM across all tumor types. In particular, myeloid cells, including macrophages and monocytes, showed high EPGM expression, indicating an active role of embryonic pro-angiogenesis pathways in tumors. A subset analysis revealed 20 distinct myeloid subtypes with varying EPGM and TPGM expression across different cancers. Treatment and disease stage influenced these gene expressions, with certain subtypes, such as HSPAhi/STAT1+ macrophages in breast cancer, displaying reduced pro-angiogenic gene activity post-treatment.
Conclusion
This study provides evidence that tumors may exploit EPGM to enhance vascularization and support sustained growth, as evidenced by the elevated EPGM expression in tumor-associated myeloid cells. The consistent presence of EPGM in TAMs across multiple cancer types suggests a conserved mechanism wherein tumors harness embryonic angiogenic pathways to facilitate their progression. Distinct EPGM expression patterns in specific myeloid cell subsets indicate potential therapeutic targets, particularly in cases where EPGM activation contributes to resistance against anti-angiogenic therapies. These findings shed new light on the molecular mechanisms underlying tumor angiogenesis and highlight the prognostic relevance of EPGM expression in cancer, underscoring its potential as a biomarker for clinical applications.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


