Design, Synthesis, and Biological Evaluation of Novel Diaminopyrimidine Macrocycles as Fourth Generation Reversible EGFR Inhibitors That Overcome Clinical Resistance to Osimertinib Mediated by C797S Mutation
IF 6.8
1区 医学
Q1 CHEMISTRY, MEDICINAL
引用次数: 0
Abstract
Overcoming clinical resistance to osimertinib mediated by the tertiary C797S mutation remains an unmet medical need. To date, there are no effective drugs that have been approved for patients who harbor EGFRT790M/C797S mutations. Herein, we applied a structure-based drug design strategy to discover a series of potent and selective diaminopyrimidine macrocycles as novel EGFRT790M/C797S inhibitors. The representative compound 21v potently inhibited EGFR19del/T790M/C797S and EGFRL858R/T790M/C797S mutants with IC50 values of 2.3 nM and 12.5 nM, respectively, and exhibited antiproliferative activity against Ba/F3-EGFR19del/T790M/C797S and Ba/F3-EGFRL858R/T790M/C797S cells with IC50 values of 41 and 52 nM, respectively. Further, 21v inhibited proliferation of the EGFR19del/T790M/C797S mutant PC-9-OR NSCLC cell line with an IC50 value of 56 nM and displayed selectivity over parental Ba/F3 and A431 cells. Moreover, 21v exhibited antitumor efficacy in a Ba/F3-EGFR19del/T790M/C797S xenograft model. This study provides a promising macrocyclic lead for anticancer drug discovery overcoming EGFRC797S mutation mediated resistance in NSCLC patients.
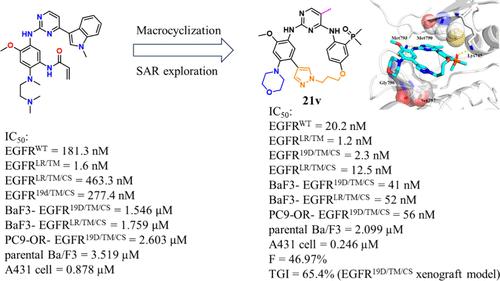
新型二氨基嘧啶大环作为第四代可逆表皮生长因子受体(EGFR)抑制剂的设计、合成和生物学评价,可克服 C797S 突变介导的奥希替尼临床耐药性
克服由三级 C797S 突变介导的奥希替尼临床耐药性仍是一项尚未满足的医疗需求。迄今为止,还没有针对 EGFRT790M/C797S 突变患者的有效药物获得批准。在此,我们采用基于结构的药物设计策略,发现了一系列作为新型 EGFRT790M/C797S 抑制剂的强效、选择性二氨基嘧啶大环。代表性化合物21v对EGFR19del/T790M/C797S和EGFRL858R/T790M/C797S突变体具有强效抑制作用,IC50值分别为2.3 nM和12.5 nM;对Ba/F3-EGFR19del/T790M/C797S和Ba/F3-EGFRL858R/T790M/C797S细胞具有抗增殖活性,IC50值分别为41 nM和52 nM。此外,21v 还能抑制 EGFR19del/T790M/C797S 突变体 PC-9-OR NSCLC 细胞系的增殖,IC50 值为 56 nM,并对亲本 Ba/F3 和 A431 细胞具有选择性。此外,21v 在 Ba/F3-EGFR19del/T790M/C797S 异种移植模型中表现出抗肿瘤疗效。这项研究为克服 NSCLC 患者中 EGFRC797S 突变介导的耐药性提供了一种很有前景的大环化合物。
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文
来源期刊

Journal of Medicinal Chemistry
医学-医药化学
CiteScore
4.00
自引率
11.00%
发文量
804
审稿时长
1.9 months
期刊介绍:
The Journal of Medicinal Chemistry is a prestigious biweekly peer-reviewed publication that focuses on the multifaceted field of medicinal chemistry. Since its inception in 1959 as the Journal of Medicinal and Pharmaceutical Chemistry, it has evolved to become a cornerstone in the dissemination of research findings related to the design, synthesis, and development of therapeutic agents.
The Journal of Medicinal Chemistry is recognized for its significant impact in the scientific community, as evidenced by its 2022 impact factor of 7.3. This metric reflects the journal's influence and the importance of its content in shaping the future of drug discovery and development. The journal serves as a vital resource for chemists, pharmacologists, and other researchers interested in the molecular mechanisms of drug action and the optimization of therapeutic compounds.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


