Bee venom and melittin: Potent key enzyme inhibitors with promising therapeutic potential
IF 2.6
4区 医学
Q2 PHARMACOLOGY & PHARMACY
引用次数: 0
Abstract
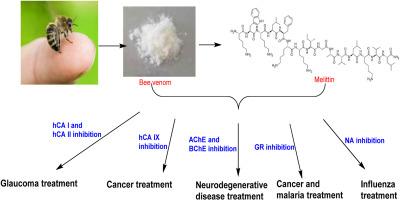
Bee venom (BV) is a versatile product with extensive applications, boasting antibacterial and anticancer properties. Within this study, we focused on isolating melittin (Mel) from Apis mellifera L. venom and exploring the influence of both BV and Mel on specific enzymes, namely carbonic anhydrase (CA) I, CA II, CA IX, glutathione reductase (GR), acetylcholinesterase (AChE), butyrylcholinesterase (BChE), and neuraminidase (NA). The rationale for selecting these enzymes is that their inhibitors have a particular interest in medicinal chemistry in the treatment of several diseases. BV was obtained using a poison collection apparatus, and Mel was isolated by means of High-Performance Liquid Chromatography (HPLC). All enzymes, except for CA I and CA II, were commercially sourced and of high purity, and the enzyme assays were carried out spectrophotometrically.
Our findings showed that BV inhibited the enzymes with IC50 values of 0.583–3.32 ng/mL, and Mel showed an inhibition range of 0.528–3.2 ng/mL. These results underscore the potential therapeutic promise of BV and Mel as robust enzyme inhibitors, offering prospects for addressing diverse health conditions.
蜂毒和美乐汀:具有治疗潜力的强效关键酶抑制剂。
蜂毒(BV)是一种用途广泛的产品,具有抗菌和抗癌特性。在这项研究中,我们重点从蜜蜂毒液中分离 Melittin(Mel),并探索 BV 和 Mel 对特定酶的影响,即碳酸酐酶 (CA) I、CA II、CA IX、谷胱甘肽还原酶 (GR)、乙酰胆碱酯酶 (AChE)、丁酰胆碱酯酶 (BChE) 和神经氨酸酶 (NA)。之所以选择这些酶,是因为它们的抑制剂在治疗多种疾病的药物化学中具有特殊的意义。BV 是通过毒物收集装置获得的,而 Mel 则是通过高效液相色谱法(HPLC)分离的。除 CA I 和 CA II 外,所有酶的来源均为高纯度的商用酶,酶测定采用分光光度法进行。我们的研究结果表明,BV 对酶的抑制作用 IC50 值为 0.583-3.32 纳克/毫升,而 Mel 的抑制作用范围为 0.528-3.2 纳克/毫升。这些结果凸显了 BV 和 Mel 作为强效酶抑制剂的潜在治疗前景,为解决各种健康问题提供了可能。
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文
来源期刊

Toxicon
医学-毒理学
CiteScore
4.80
自引率
10.70%
发文量
358
审稿时长
68 days
期刊介绍:
Toxicon has an open access mirror Toxicon: X, sharing the same aims and scope, editorial team, submission system and rigorous peer review. An introductory offer Toxicon: X - full waiver of the Open Access fee.
Toxicon''s "aims and scope" are to publish:
-articles containing the results of original research on problems related to toxins derived from animals, plants and microorganisms
-papers on novel findings related to the chemical, pharmacological, toxicological, and immunological properties of natural toxins
-molecular biological studies of toxins and other genes from poisonous and venomous organisms that advance understanding of the role or function of toxins
-clinical observations on poisoning and envenoming where a new therapeutic principle has been proposed or a decidedly superior clinical result has been obtained.
-material on the use of toxins as tools in studying biological processes and material on subjects related to venom and antivenom problems.
-articles on the translational application of toxins, for example as drugs and insecticides
-epidemiological studies on envenoming or poisoning, so long as they highlight a previously unrecognised medical problem or provide insight into the prevention or medical treatment of envenoming or poisoning. Retrospective surveys of hospital records, especially those lacking species identification, will not be considered for publication. Properly designed prospective community-based surveys are strongly encouraged.
-articles describing well-known activities of venoms, such as antibacterial, anticancer, and analgesic activities of arachnid venoms, without any attempt to define the mechanism of action or purify the active component, will not be considered for publication in Toxicon.
-review articles on problems related to toxinology.
To encourage the exchange of ideas, sections of the journal may be devoted to Short Communications, Letters to the Editor and activities of the affiliated societies.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


