Deciphering the molecular mechanism underlying morphology transition in two-component DNA-protein cophase separation
IF 4.4
2区 生物学
Q2 BIOCHEMISTRY & MOLECULAR BIOLOGY
引用次数: 0
Abstract
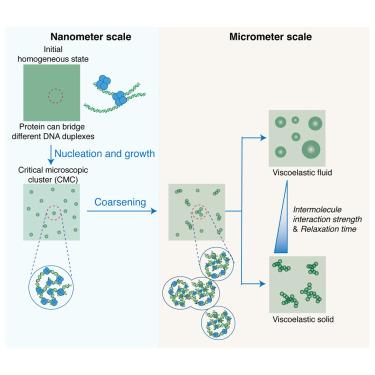
Nucleic acid and protein co-condensates exhibit diverse morphologies crucial for fundamental cellular processes. Despite many previous studies that advanced our understanding of this topic, several interesting biophysical questions regarding the underlying molecular mechanisms remain. We investigated DNA and human transcription factor p53 co-condensates—a scenario where neither dsDNA nor the protein demonstrates phase-separation behavior individually. Through a combination of experimental assays and theoretical approaches, we elucidated: (1) the phase diagram of DNA-protein co-condensates at a certain observation time, identifying a phase transition between viscoelastic fluid and viscoelastic solid states, and a morphology transition from droplet-like to “pearl chain”-like co-condensates; (2) the growth dynamics of co-condensates. Droplet-like and “pearl chain”-like co-condensates share a common initial critical microscopic cluster size at the nanometer scale during the early stage of phase separation. These findings provide important insights into the biophysical mechanisms underlying multi-component phase separation within cellular environments.
破译双组分 DNA 蛋白共相分离中形态转变的分子机制
核酸和蛋白质共缩合物呈现出对基本细胞过程至关重要的各种形态。尽管之前的许多研究加深了我们对这一主题的理解,但有关其基本分子机制的几个有趣的生物物理问题依然存在。我们研究了 DNA 和人类转录因子 p53 的共缩合物--在这种情况下,dsDNA 和蛋白质都不会单独表现出相分离行为。通过实验和理论相结合的方法,我们阐明了:(1)DNA-蛋白质共凝物在一定观察时间内的相图,确定了粘弹性流体态和粘弹性固态之间的相变,以及从液滴状共凝物到 "珍珠链 "状共凝物的形态转变;(2)共凝物的生长动力学。在相分离的早期阶段,"液滴状 "和 "珍珠链状 "共凝聚物在纳米尺度上具有共同的初始临界微观团簇尺寸。这些发现为了解细胞环境中多组分相分离的生物物理机制提供了重要的启示。
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文
来源期刊

Structure
生物-生化与分子生物学
CiteScore
8.90
自引率
1.80%
发文量
155
审稿时长
3-8 weeks
期刊介绍:
Structure aims to publish papers of exceptional interest in the field of structural biology. The journal strives to be essential reading for structural biologists, as well as biologists and biochemists that are interested in macromolecular structure and function. Structure strongly encourages the submission of manuscripts that present structural and molecular insights into biological function and mechanism. Other reports that address fundamental questions in structural biology, such as structure-based examinations of protein evolution, folding, and/or design, will also be considered. We will consider the application of any method, experimental or computational, at high or low resolution, to conduct structural investigations, as long as the method is appropriate for the biological, functional, and mechanistic question(s) being addressed. Likewise, reports describing single-molecule analysis of biological mechanisms are welcome.
In general, the editors encourage submission of experimental structural studies that are enriched by an analysis of structure-activity relationships and will not consider studies that solely report structural information unless the structure or analysis is of exceptional and broad interest. Studies reporting only homology models, de novo models, or molecular dynamics simulations are also discouraged unless the models are informed by or validated by novel experimental data; rationalization of a large body of existing experimental evidence and making testable predictions based on a model or simulation is often not considered sufficient.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


