6π 1,5-Diaza-electrocyclization of Isatin, α-Ketoester Derived Hydrazones with α,β-Unsaturated Ketoesters, Ketones, and Aldehydes Enables Functionalized Pyrazoles
IF 3.3
2区 化学
Q1 CHEMISTRY, ORGANIC
引用次数: 0
Abstract
A novel and efficient 6π 1,5-diaza-electrocyclization of readily available isatin, α-ketoester derived hydrazones with α,β-unsaturated ketoesters, ketones, and aldehydes has been developed. A series of functionalized pyrazoles bearing an oxindole unit were easily afforded by this tool with good yields and high functional group tolerance under mild conditions. Importantly, the obtained pyrazole could be further converted to 3-pyrazolecarboxylic acid with a quaternary carbon stereocenter.
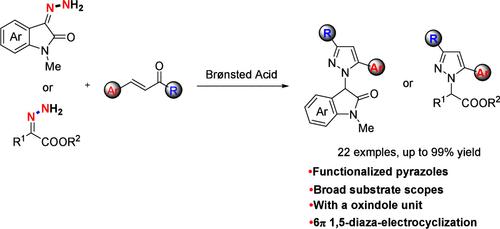
6π 1,5-二氮杂环化伊沙廷、α-酮甾烷衍生肼与α,β-不饱和酮甾烷、酮和醛实现功能化吡唑
本研究开发了一种新颖高效的 6π 1,5-二氮电环化方法,可将现成的异汀、α-酮衍生的肼与α,β-不饱和酮、酮和醛进行电环化。在温和的条件下,利用这种工具很容易得到一系列带有吲哚单元的官能化吡唑,而且产量高、官能团耐受性强。重要的是,所获得的吡唑可进一步转化为具有季碳立体中心的 3-吡唑羧酸。
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文
来源期刊

The Journal of Organic Chemistry
化学-有机化学
CiteScore
6.20
自引率
11.10%
发文量
1467
审稿时长
2 months
期刊介绍:
The Journal of Organic Chemistry welcomes original contributions of fundamental research in all branches of the theory and practice of organic chemistry. In selecting manuscripts for publication, the editors place emphasis on the quality and novelty of the work, as well as the breadth of interest to the organic chemistry community.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


