Effects of A’-Site Cation Structure on the Stability of 2D Tin Halide Perovskites
IF 7.2
2区 材料科学
Q2 CHEMISTRY, PHYSICAL
引用次数: 0
Abstract
Two-dimensional halide perovskites (2D-HPs) are of significant interest for their applications in optoelectronic devices. Part of this increased interest in 2D-HPs stems from their increased stability relative to their three-dimensional (3D) counterparts. Here, the origin of higher stability in 2D-HPs is mainly attributed to the bulky ammonium cation layers, which can act as a blocking layer against moisture and oxygen ingression and ion diffusion. While 2D-HPs have demonstrated increased stability, it is not clear how the structure of the ammonium ions impacts material stability. Herein, we investigate how the structure of ammonium cations, including three n-alkyl ammoniums, phenethylammonium (PEA) and five PEA derivatives, anilinium (An), benzylammonium (BzA), and cyclohexylmethylammonium (CHMA), affects the crystal structure and air, water, and oxygen stability of 2D tin halide perovskites (2D-SnHPs). We find that stability is influenced by several factors, including the molecular packing and intermolecular interactions in the organic layer, steric effects around the ammonium group, the orientation distribution of the 2D sheets, and the hydrophobicity of the perovskite film surface. With superior hydrophobicity, strong interactions between organic molecules, and a high extent of parallel oriented inorganic sheets, the 2-(4-trifluoromethyl-phenyl)-ethylammonium (4-TFMPEA) ion forms the most stable 2D-SnHP among the 12 ammonium cations investigated.
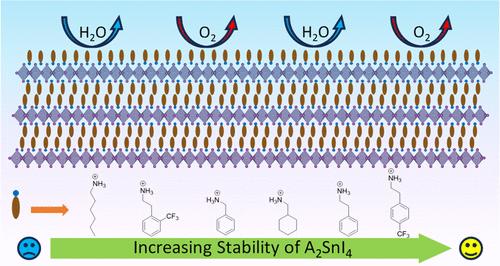
A'-Site 阳离子结构对二维卤化锡包晶石稳定性的影响
二维卤化物包晶(2D-HPs)因其在光电设备中的应用而备受关注。人们对二维卤化物包光体的兴趣日益浓厚,部分原因是二维卤化物包光体与三维卤化物包光体相比稳定性更高。在这里,2D-HPs 更高的稳定性主要归因于其庞大的铵阳离子层,它可以作为阻挡层,防止水分和氧气的侵入以及离子的扩散。虽然 2D-HPs 显示出更高的稳定性,但还不清楚铵离子的结构如何影响材料的稳定性。在此,我们研究了铵阳离子(包括三种正烷基铵、苯乙基铵(PEA)和五种 PEA 衍生物、苯胺(An)、苄基铵(BzA)和环己基甲基铵(CHMA))的结构如何影响二维卤化锡包晶石(2D-SnHPs)的晶体结构以及空气、水和氧的稳定性。我们发现稳定性受多个因素的影响,包括有机层中的分子堆积和分子间相互作用、铵基团周围的立体效应、二维片的取向分布以及包晶薄膜表面的疏水性。2-(4-trifluoromethyl-phenyl)-ethylammonium (4-TFMPEA) 离子具有优异的疏水性、有机分子间的强相互作用和高度平行取向的无机薄片,在所研究的 12 个铵阳离子中形成了最稳定的二维-SnHP。
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文
来源期刊

Chemistry of Materials
工程技术-材料科学:综合
CiteScore
14.10
自引率
5.80%
发文量
929
审稿时长
1.5 months
期刊介绍:
The journal Chemistry of Materials focuses on publishing original research at the intersection of materials science and chemistry. The studies published in the journal involve chemistry as a prominent component and explore topics such as the design, synthesis, characterization, processing, understanding, and application of functional or potentially functional materials. The journal covers various areas of interest, including inorganic and organic solid-state chemistry, nanomaterials, biomaterials, thin films and polymers, and composite/hybrid materials. The journal particularly seeks papers that highlight the creation or development of innovative materials with novel optical, electrical, magnetic, catalytic, or mechanical properties. It is essential that manuscripts on these topics have a primary focus on the chemistry of materials and represent a significant advancement compared to prior research. Before external reviews are sought, submitted manuscripts undergo a review process by a minimum of two editors to ensure their appropriateness for the journal and the presence of sufficient evidence of a significant advance that will be of broad interest to the materials chemistry community.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


