Development of hybrid aptamers-engineered PROTACs for degrading VEGF165 in both tumor- and vascular endothelial cells
IF 6
2区 医学
Q1 CHEMISTRY, MEDICINAL
引用次数: 0
Abstract
Tumors and angiogenesis are connected through a complex interplay. VEGF165, generated from both tumor and vascular endothelial cells, serves as a mutual benefit for both cell types. Therapeutic approaches modulating VEGF165 have been proposed as promising antitumor therapies. PROTACs are bifunctional molecules that exploit the intracellular ubiquitin-proteasome system to degrade specific proteins. To date, there are no targeted PROTACs designed to degrade VEGF165 in both tumor and vascular endothelial cells. The aptamer AS1411 is notable for its ability to selectively recognize and enter both tumor and vascular endothelial cells by targeting the cell surface nucleolin (NCL). Moreover, AS1411 has also been repurposed as an intracellular recruiter of E3 ligase MDM2 via leveraging NCL as a molecular bridge. In this study, we conjugated AS1411 with a VEGF165-specific aptamer V7t1, creating hybrid aptamers-engineered PROTACs. The PROTACs demonstrate remarkable selectivity for both tumor and vascular endothelial cells and facilitate the ubiquitination and proteasomal degradation of VEGF165. The PROTACs inhibit the growth of tumor cells and also impede angiogenesis, without causing toxicity to normal tissues. The hybrid aptamers-engineered PROTACs provide an avenue for disrupting the tumor-angiogenesis interplay through modulation of VEGF165 in both tumor and vascular endothelial cells.
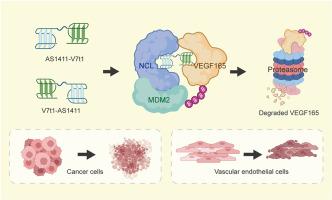
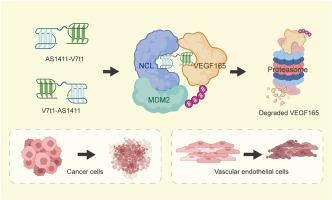
开发可在肿瘤细胞和血管内皮细胞中降解 VEGF165 的混合适配体工程化 PROTACs
肿瘤和血管生成通过复杂的相互作用联系在一起。由肿瘤细胞和血管内皮细胞产生的 VEGF165 对两种细胞类型都有好处。调控 VEGF165 的治疗方法被认为是有前景的抗肿瘤疗法。PROTACs 是一种双功能分子,可利用细胞内泛素-蛋白酶体系统降解特定蛋白质。迄今为止,还没有设计出同时降解肿瘤细胞和血管内皮细胞中 VEGF165 的靶向 PROTACs。适配体 AS1411 的显著特点是能通过靶向细胞表面的核素蛋白(NCL),选择性地识别并进入肿瘤细胞和血管内皮细胞。此外,AS1411 还通过利用 NCL 作为分子桥,被重新用作 E3 连接酶 MDM2 的细胞内招募剂。在本研究中,我们将 AS1411 与 VEGF165 特异性拟合物 V7t1 共轭,创造出混合拟合物工程化的 PROTACs。PROTACs 对肿瘤细胞和血管内皮细胞都具有显著的选择性,能促进 VEGF165 的泛素化和蛋白酶体降解。PROTACs 可抑制肿瘤细胞的生长,阻碍血管生成,但不会对正常组织造成毒性。这种由混合适配体设计的 PROTACs 为通过调节肿瘤细胞和血管内皮细胞中的 VEGF165 来破坏肿瘤与血管生成之间的相互作用提供了一条途径。
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文
来源期刊
CiteScore
11.70
自引率
9.00%
发文量
863
审稿时长
29 days
期刊介绍:
The European Journal of Medicinal Chemistry is a global journal that publishes studies on all aspects of medicinal chemistry. It provides a medium for publication of original papers and also welcomes critical review papers.
A typical paper would report on the organic synthesis, characterization and pharmacological evaluation of compounds. Other topics of interest are drug design, QSAR, molecular modeling, drug-receptor interactions, molecular aspects of drug metabolism, prodrug synthesis and drug targeting. The journal expects manuscripts to present the rational for a study, provide insight into the design of compounds or understanding of mechanism, or clarify the targets.

 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


