Integrated proteomics and transcriptomics analysis reveals insights into differences in premature mortality associated with disparate pathogenic RBM20 variants
IF 4.9
2区 医学
Q1 CARDIAC & CARDIOVASCULAR SYSTEMS
引用次数: 0
Abstract
Variants in RNA binding motif protein 20 (RBM20) are causative in a severe form of dilated cardiomyopathy referred to as RBM20 cardiomyopathy, yet the mechanisms are unclear. Moreover, the reason(s) for phenotypic heterogeneity in carriers with different pathogenic variants are similarly opaque. To gain insight, we carried out multi-omics analysis, including the first analysis of gene expression changes at the protein level, of mice carrying two different pathogenic variants in the RBM20 nuclear localization signal (NLS). Direct comparison of the phenotypes confirmed greater premature morality in S639G variant carrying mice compared to mice with the S637A variant despite similar cardiac remodeling and dysfunction. Analysis of differentially spliced genes uncovered alterations in the splicing of both RBM20 target genes and non-target genes, including several genes previously implicated in arrhythmia. Global proteomics analysis found that a greater number of proteins were differentially expressed in the hearts of Rbm20S639G mice relative to WT than in Rbm20S637A versus WT. Gene ontology analysis suggested greater mitochondrial dysfunction in Rbm20S639G mice, although direct comparison of protein expression in the hearts of Rbm20S639G versus Rbm20S637A mice failed to identify any significant differences. Similarly, few differences were found by direct comparison of gene expression at the transcript level in Rbm20S639G and Rbm20S637A despite greater coverage. Our data provide a comprehensive overview of gene splicing and expression differences associated with pathogenic variants in RBM20, as well as insights into the molecular underpinnings of phenotypic heterogeneity associated with different dilated cardiomyopathy-associated variants.
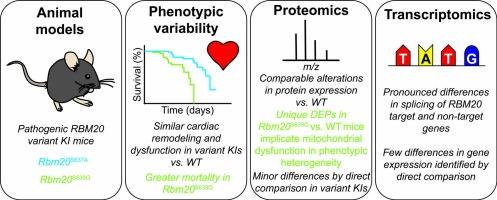
蛋白质组学和转录组学综合分析揭示了与不同致病基因 RBM20 变体相关的过早死亡率差异
RNA 结合基调蛋白 20(RBM20)变异是一种严重扩张型心肌病(称为 RBM20 心肌病)的致病因子,但其机制尚不清楚。此外,不同致病变体携带者表型异质性的原因也同样不清楚。为了深入了解这一问题,我们对携带 RBM20 核定位信号(NLS)两种不同致病变体的小鼠进行了多组学分析,包括首次分析蛋白质水平的基因表达变化。表型的直接比较证实,与携带 S637A 变异的小鼠相比,携带 S639G 变异的小鼠过早死亡率更高,尽管心脏重塑和功能障碍相似。对不同剪接基因的分析发现,RBM20靶基因和非靶基因的剪接都发生了改变,其中包括几个以前与心律失常有关的基因。全局蛋白质组学分析发现,Rbm20S639G 小鼠心脏中相对于 WT 有差异表达的蛋白质数量多于 Rbm20S637A 相对于 WT。基因本体分析表明,Rbm20S639G 小鼠的线粒体功能障碍更严重,但直接比较 Rbm20S639G 与 Rbm20S637A 小鼠心脏中的蛋白质表达,未能发现任何显著差异。同样,通过直接比较 Rbm20S639G 和 Rbm20S637A 在转录本水平的基因表达,尽管覆盖范围更大,但几乎没有发现差异。我们的数据全面概述了与 RBM20 致病变异相关的基因剪接和表达差异,并揭示了与不同扩张型心肌病相关变异相关的表型异质性的分子基础。
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文
来源期刊
CiteScore
10.70
自引率
0.00%
发文量
171
审稿时长
42 days
期刊介绍:
The Journal of Molecular and Cellular Cardiology publishes work advancing knowledge of the mechanisms responsible for both normal and diseased cardiovascular function. To this end papers are published in all relevant areas. These include (but are not limited to): structural biology; genetics; proteomics; morphology; stem cells; molecular biology; metabolism; biophysics; bioengineering; computational modeling and systems analysis; electrophysiology; pharmacology and physiology. Papers are encouraged with both basic and translational approaches. The journal is directed not only to basic scientists but also to clinical cardiologists who wish to follow the rapidly advancing frontiers of basic knowledge of the heart and circulation.

 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


